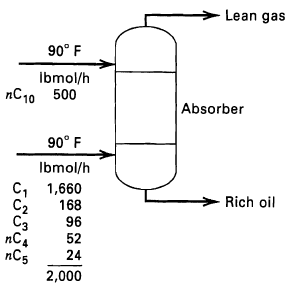
Determine by the Kremser method the separation that can be achieved for the absorption operation indicated in
Question:
Determine by the Kremser method the separation that can be achieved for the absorption operation indicated in Figure for the following combinations of conditions:
(a) Six equilibrium stages and 75 psia operating pressure,
(b) Three equilibrium stages and 150 psia operating pressure,
(c) Six equilibrium stages and 150 psia operating pressure. At 90°F and 75 psia, the K-value of nC10 =0.0011.
Transcribed Image Text:
Lean gas 90° F Ibmol/h пС10 500 Absorber 90° F Ibmol/h C, 1,660 C2 Сз пC nC5 Rich oil 168 96 52 24 2,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Use the Kremser method with Eqs 548 for fraction not absorbed A 550 for fr...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Determine by the Kremser group method the separation that can be achieved for the absorption operation indicated in figure for the following combinations of conditions: (a) Six equilibrium stages and...
-
What limits the data rates that can be achieved with GPRS and HSCSD using real devices (compared to the theoretical limit in a GSM system)?
-
Discuss the balance that can be achieved between fixed salary, bonuses and incentives and the link with motivation of salespeople.
-
A baseball player usually has four at bats each game. Suppose the baseball player is a lifetime 0.25 hitter. Find the probability that this player will have: (a) Two hits out of four at bats (b) No...
-
Data for Kicked-Back Tennis Shop are presented in P5-4B. In P5-4B At the beginning of the current tennis season, on April 1, 2012, the general ledger of Kicked-Back Tennis Shop showed Cash $4,000;...
-
Compare and contrast multi-domestic, global, and transnational strategies. What environmental factors influence a firm's international strategy? What are the benefits and risks of international...
-
A \(3 \mathrm{ft} \times 5 \mathrm{ft}\) double-domed skylight has outer and inner fat-plate plastic transmittances of 0.8 and 0.7, respectively; a \(2 \mathrm{ft}\) deep well with \(80 \%\)...
-
The general ledger of jackrabbit Rentals at January 1, 2018, includes the following account balances: The following is a summary of the transactions for the year: a. January 12Provide services to...
-
A residential development of 280-house is being planned. Assumethat American Water Works Association (AWWA) average dailyhousehold consumption applies and that each house has 3 residents.Estimate the...
-
The balance sheet of Fern Ltd. appears below: Fern Ltd. Balance Sheet December 31, 2022 Cash Accounts Receivables Inventory Net Fixed Assets Total Assets Accruals Accounts Payable Short term debt...
-
Sulfur dioxide and butadienes (B3 and B2) are to be stripped with nitrogen from the liquid stream as shown in Figure so that butadiene sulfone (BS) product will contain less than 0.05 mol% SO2 and...
-
One thousand kilomoles per hour of rich gas at 70F with 25% C1, 15% C2,25% C3, 20% nC4, and 15% nC5 by moles is to be absorbed by 500 kmol/h of nC10 at 90F in an absorber operating at 4 atm....
-
Using a thermometer, measure the boiling temperature of water and calculate the corresponding saturation pressure. From this information, estimate the altitude of your town and compare it with the...
-
A drummer engaged to play for a pop group was contractually bound to work seven nights a week. After falling ill, Jacks doctor advised he work only four nights a week. It became necessary for the pop...
-
How can you determine whether an organism is heterozygous or homozygous for a dominant trait?
-
A company has employed an independent contractor to construct a reservoir on its land. There were old disused mining shafts under the reservoir site and they led to an adjoining coal mine. The shafts...
-
As mentioned in the chapter, prior experience in an industry helps entrepreneurs recognize business opportunities. This concept extends to prior experience in any aspect of lifewhether it is in...
-
Rawlsworth is an employee of General Sam Corporation. One of Rawlsworths jobs is to monitor the amount of particular pollutants and to record the results on a form that is submitted to the...
-
The HRCI Exam Content Outline Website https://www.hrci.org/docs/defaultsource/web-files/phr-exam-content-outline.pdf ?sfvrsn=13c44f61_28 lists the knowledge someone studying for the HRCI PHR...
-
Calculate the electrical conductivity of a fiber-reinforced polyethylene part that is reinforced with 20 vol % of continuous, aligned nickel fibers.
-
Discuss the various alternative forms of international assignment.
-
At the particle settling velocity, what force balances the drag force plus the buoyant force?
-
For a solid, spherical particle of 0.8 mm in diameter and a density of 2,600 kg/m 3 that is immersed in a fluid of density 1,200 kg/ m 3 and a viscosity of 1.0 cP, calculate (a) The unhindered...
-
Why is particle size the main parameter used in selecting a mechanical phase-separation device?
-
Simplify. -3(a + b[2a-a(4 - b)])
-
Defining strategy is not simple. Understanding different ways of thinking about strategy is the first toward mastering the art and science of strategic management. Strategy is a complex concept that...
-
The following is a list of 13 measurements. -61, 75, -92, -11, 61, 27, -13, -54, 34, 66, 96, 30, -64 Send data to calculator Suppose that these 13 measurements are respectively labeled X1, X2, X13....

Study smarter with the SolutionInn App


