Determine the diameter and packed height of a countercurrently operated packed tower required to recover 99% of
Question:
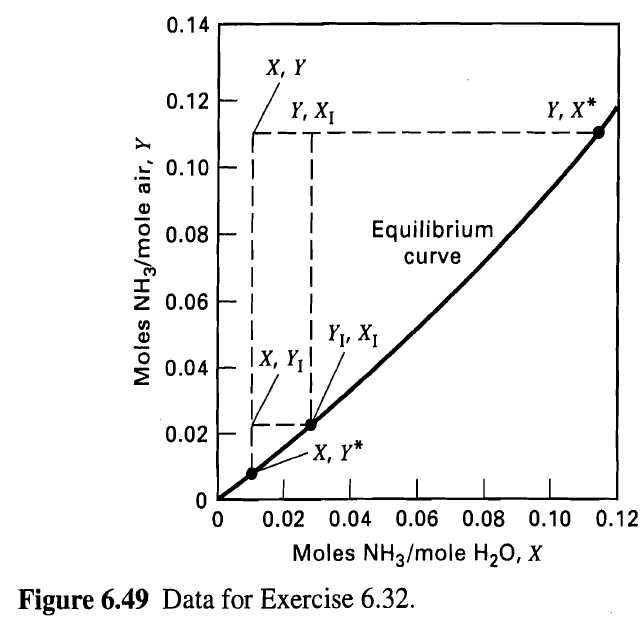
Determine the diameter and packed height of a countercurrently operated packed tower required to recover 99% of the ammonia from a gas mixture that contains 6 mol% NH3 in air. The tower, packed with l-in. metal Pall rings, must handle 2,000 ft3/min of gas as measured at 68oF and 1 atm. The entering water-absorbent rate will be twice the theoretical minimum, and the gas velocity will be such that it is 50% of the flooding velocity. Assume isothermal operation at 68°F and 1 atm. Equilibrium data are given in Figure 6.49
Transcribed Image Text:
0.14 X, Y 0.12 Y, X* Y, X1 0.10 | Equilibrium 0.08 curve 0.06 EI Y1, X1 |X, Y¡! 0.04 0.02 -х, ү* 0.02 0.04 0.06 0.08 0.10 0.12 Moles NH3/mole H20, X Figure 6.49 Data for Exercise 6.32. Moles NH3/mole air, Y
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Entering gas rate is Compute material balance NH 3 in entering gas 006312 1872 lbmolh NH 3 in exiting gas 0011872 019 lbmolh Air in entering and exiti...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A rigid 35 ft3 tank contains water initially at 250 F, with 50 % liquid and 50% vapor, by volume. A pressure-relief valve on the top of the tank is set to 150 lbf/in 2 (the tank pressure cannot...
-
A mixture of N2, H2 and NH3 is at equilibrium according to the equation N2(g) + 3H2(g) 2NH3(g) as depicted below. The volume is suddenly decreased (by increasing the external pressure), and a new...
-
A rigid tank contains 1 lb mol of argon gas at 400 R and 750 psia. A valve is now opened, and 3 lb mol of N2 gas is allowed to enter the tank at 340 R and 1200 psia. The final mixture temperature is...
-
Name four ways to mitigate the incentives for managers to overproduce.
-
Distinguish between the journal entries in a periodic inventory system and a perpetual inventory system (a) Made by the buyer, (b) Made by the seller.
-
AppleBerry (AB) is a high-tech manufacturer of digital products. Recently, it has been very successful at manufacturing a portable MP3 player, the Curvy. The Curvy comes in two forms: one with 32 GB...
-
Calculate the maximum power and LCOE for a parabolic collector similar to that in Example 8.1, but assume that an evacuated tube receiver is used (case E in Figure 8.18a). Example 8.1 Figure 8.18...
-
Mandy Knox is the new manager of the materials storeroom for Timken Manufacturing. Mandy has been asked to estimate future monthly purchase costs for part # 696, used in two of Timkens products....
-
East Company purchased West Company. East Company paid $ 6 0 0 , 0 0 0 cash and assumed all of West Company s liabilities. West's books showed tangible assets of $ 5 5 0 , 0 0 0 , liabilities of $ 4...
-
On January 1, 2020, Zulu Co. sells and immediately leases back a building from X-Ray Inc. The building has a net book value of $30,000,000 ($80,000,000 cost $50,000,000 accumulated depreciation) and...
-
You are asked to design a packed column to recover acetone from air continuously, by absorption with water at 60F. The air contains 3 mol% acetone, and a 97% recovery is desired. The gas flow rate is...
-
A tower, packed with Montz B1-200 metal structured packing, is to be designed to absorb SO2 from air by scrubbing with water. The entering gas, at an SO2-free flow rate of 6.90 lbmollh-ft2 of bed...
-
Under what conditions must a company emerging from a bankruptcy reorganization apply fresh start accounting?
-
If the outlay is lower by the amount that Simpson suggests, the project NPV should increase by an amount closest to: A. 0.09 billion. B. 0.14 billion. C. 0.17 billion. Barbara Simpson is a sell-side...
-
Steve Jackson (birthdate December 13, 1967) is a single taxpayer living at 3215 Pacific Dr., Apt. B, Pacific Beach, CA 92109. His Social Security number is 465-88-9415. In 2020, Steves earnings and...
-
A switch from straight-line to accelerated depreciation would: A. increase the NPV and decrease the first year operating income after taxes. B. increase the first year operating income after taxes...
-
Simpson should estimate the initial outlay and the terminal year nonoperating cash flow, respectively, to be closest to: A. 1.50 billion and 0.70 billion. B. 1.90 billion and 0.70 billion. C. 1.90...
-
The following are selected account balances for Warren Clinic as of December 31, 2015, in alphabetical order. Create Warren Clinics balance sheet. Accounts payable Accounts receivable, net Cash $...
-
Given what Kia UK accomplished, should Tesla implement such an engagement program? Why?
-
Why is the national security argument for tariffs questionable?
-
The basic idea behind the pseudoscientific subliminal selfhelp industry is that you can change some aspect of your behavior if you let your unconscious process motivational messages (you can lose...
-
A total of 6,000 lb/h of a liquid solution of 40 wt% benzene in naphthalene at 50 C is cooled to 15 C. Use Figure 4.22 to obtain the weight of crystals and the flow rate and composition of mother...
-
A mixture of chloroform (CHCl 3 ) and acetic acid at 18 C and 1 atm (101.3 kPa) is extracted with water to recover the acid. Fortyfive kg of 35 wt% CHCl3 and 65 wt% acid is treated with 22.75 kg of...
-
In a rigorous vaporliquidliquid equilibrium calculation (the so-called three-phase flash), is it necessary to consider all possible phase conditions, i.e., all-liquid, all-vapor, vaporliquid, liquid...
-
How can leaders effectively navigate the complexities of cross-cultural leadership to build cohesive and inclusive multinational teams ?
-
BB Co produced the COGM Schedule for the month-ending January 31, 2023. Below, certain amounts and line headings are missing from the COGM Schedule (shaded cells). Complete the COGM Schedule. Type...
-
P 4 - - Cost - Volume - Profit Relationship ( 2 / 7 ) AV Inc. has developed an A - Pod and an A - Tune which have become popular among young adults. Last year's cost and operating data for the two...

Study smarter with the SolutionInn App


