Determine the rate of change HG of the angular momentum HG of the disk of Prob. 18.4
Question:
Determine the rate of change HG of the angular momentum HG of the disk of Prob. 18.4 for an arbitrary value of β, knowing that its angular velocity ω remains constant.
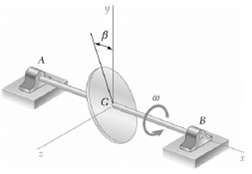
Transcribed Image Text:
B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
B Lo 17 B Use principal axes x y z as shown i cosi sin Bj j sin pi cosj i cos Bi sin ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Vector Mechanics for Engineers Statics and Dynamics
ISBN: 978-0073212227
8th Edition
Authors: Ferdinand Beer, E. Russell Johnston, Jr., Elliot Eisenberg, William Clausen, David Mazurek, Phillip Cornwell
Question Posted:
Students also viewed these Mechanical Engineering questions
-
Determine the rate of change HD of the angular momentum HD of the assembly of Prob. 18.145, assuming that at the instant considered the assembly has an angular velocity = (12 rad/s)i and an angular...
-
The velocity is equal to the rate of change of distance x (m), (a) Substitute Eq. (1.10) and develop an analytical solution for distance as a function of time. Assume that x(0) = 0. (b) Use Euler's...
-
Determine the rate of sensible heat loss from a building due to infiltration if the outdoor air at 5C and 90 kPa enters the building at a rate of 35 L/s when the indoors is maintained at 20C.
-
Is an isothermal process necessarily internally reversible? Explain your answer with an example.
-
Write a short research report explaining a recent innovation in your area of interest or expertise. Your goal is to develop a recommendation whether your company should become committed to this...
-
1. Is Worldwide Games a technology leader or a technology follower? What are the risks and benefits of staking out this position? 2. What opportunities might worldwide be missing by not having its...
-
How should a board define its optimal size? What issues influence this choice?
-
Vapor-liquid equilibrium data for mixtures of water and isopropanol at 1 atm (101.3 kPa, 760 torr) are given below. (a) Prepare T-x-y and x-y diagrams. (b) When a solution containing 40 mol %...
-
Can you Provide me a case study along with the solution in which Accounting Revenue Recognition five steps process, cash, and receivables concepts were applied as per the context of IFRS and ASPE
-
Which of the main models of international classification of accounting do you prefer? Explain your reasoning.
-
Determine the rate of change HG of the angular momentum HG of rod AB of Prob.18.3 A 12 in. 12 in. 20 B
-
Determine the rate of change HA of the angular momentum HA of the disk of Prob.18.7. - 100 200 m
-
Why are venture capital funds typically organized as limited partnerships? In particular, why are they private firms instead of public firms?
-
Assume that Corn Co. sold 7,900 units of Product A and 2,100 units of Product B during the past year. The unit contribution margins for Products A and B are $35 and $64, respectively. Corn has fixed...
-
On January 1, 2023, Hadley provided Ms. Kim with a $400,000 loan to assist with the purchase of a new home. The interest rate on the loan was 0.75% and monthly interest was withheld from each...
-
The inverse aggregate demand for energy is given by pt = 15 - wt, where wt = zt+Yt, zt is supply of the backstop, and Yt is supply of a non-renewable resource. z and y are perfect substitutes....
-
On December 31, 2026, Pink issued 50,000 shares of its common stock with a fair value of $35 per share for the net assets of Black. The following facts applied: Black dissolves immediately after the...
-
Gunlock Chair Co. is an elite manufacturer of mid-century modern solid oak chairs. At the start of May, the following budgeted unit amounts related to its manufacture of these exclusive oak chairs:...
-
Determine the deflection of the beam at its center caused by shear. The shear modulus is \(G\). I 27 - 22
-
What kind of rays are X-rays?
-
What methods are used to control neocolonies?
-
Argon gas enters a turbine at 1000 psia and 1000 R with a velocity of 300 ft/s and leaves at 150 psia and 500 R with a velocity of 450 ft/s at a rate of 12 lbm/s. Heat is being lost to the...
-
An adiabatic 0.2-m3 storage tank that is initially evacuated is connected to a supply line that carries nitrogen at 225 K and 10 MPa. A valve is opened, and nitrogen flows into the tank from the...
-
For a homogeneous (single-phase) simple pure substance, the pressure and temperature are independent properties, and any property can be expressed as a function of these two properties. Taking v = v...
-
I can't calculate the option AB and theNPV how to do
-
June 30, 2021, the market interest rate is 9%. Ramsey Corporation issues $550,000 of 12%, 20-year bonds payable. The bonds pay interest on June 30 and December 31. The company amortizes bond premium...
-
Down Under Products sales budget for the next four months is as follows: Unit Sales April 88,000 May 90,000 June 128,000 July 99,000 The company wants its ending inventory levels to equal 15% of the...

Study smarter with the SolutionInn App


