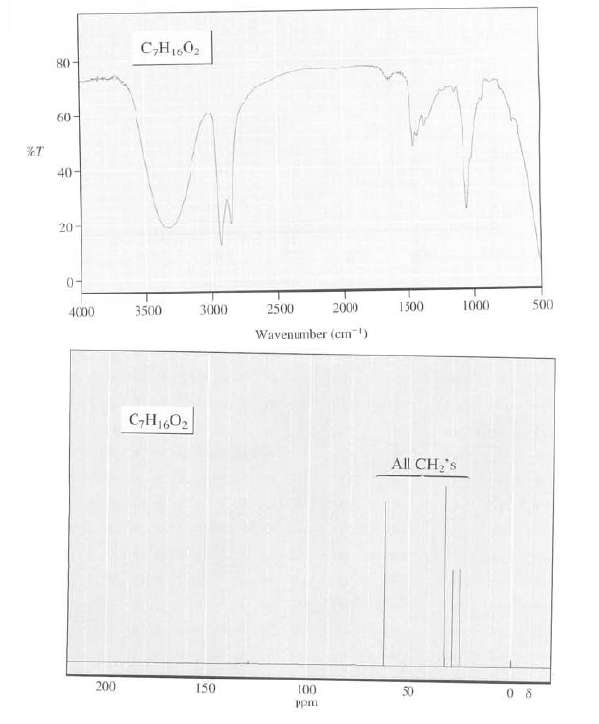
Determine the structure of this compound from its IR and 13C-NMR spectra, its formula isC7H16O2: C,H1,0, 80-
Question:
Determine the structure of this compound from its IR and 13C-NMR spectra, its formula isC7H16O2:
Transcribed Image Text:
C,H1,0, 80- 60- %T 40 20- 0- 500 1000 1500 2000 2500 3500 3000 4000 Wavenumber (cm) C,H1602 All CH's 200 150 100 50 udd
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The IR spectrum of this compound shows as OH band in the 35003...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H and 13C NMR spectra of compound A, C8H9Br are shown. Propose a structure for A, and assign peaks in the spectra to your structure. TMS O ppm 10 8. 6. Chemical shift (8) TMS 200 180 160 140 120...
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
Determine the structure of an alcohol with molecular formula C 6 H 14 O that exhibits the following DEPT-135 spectrum: DEPT-135 70 60 50 40 Chemical Shift (ppm) 30 20 10
-
All of the following are advantages of licensing EXCEPT: the foreign country gains employment by having the product manufactured locally. the licensee gains information that allows it to start with a...
-
Machiavellian describes negative character traits that include manipulation cunning, duplicity, deception, and bad faith. A Machiavellian ("Mach") rating score was determined for each in a random...
-
What might be some disadvantages of acqui-hiring? As a manager, how would you draw up a contract with a new employee who was an acqui-hire? Include considerations such as compensation and benefits,...
-
For each of the following sets of numbers, calculate a \(95 \%\) confidence interval for the mean ( \(\sigma\) not known); before going through the steps in calculating the confidence interval, the...
-
Dakota Company experienced the following events during 2018: 1. Acquired $30,000 cash from the issue of common stock. 2. Paid $12,000 cash to purchase land. 3. Borrowed $10,000 cash. 4. Provided...
-
Assume that ABC Insurance Company has purchased from QS Reinsurance Company a quota share treaty with a $500,000 limit and a retention of 30 percent and a cession of 70 percent. ABC has written...
-
Abby Ellen opened Abbys Toy House. As her newly hired accountant, your tasks are to do the following: 1. Journalize the transactions for the month of March. Abby uses special journals for sales on...
-
Assign the absorptions in the 13C-NMR spectra of these compounds to the appropriate carbons: (a) 1-Butanol; absorptions at 61.4, 35.0, 19.1, and 13.6 (b) Cyclohexanone; absorptions at 209.7, 41.9,...
-
Predict the multiplicities of the indicated hydrogen's in the 1H-NMR spectra of these compounds: CH,CI I H I - + CH,CI CI - c) b) --- I H I - ()
-
Required: In assessing the risk of material misstatement in the financial statements as a whole, why should auditors pay particular attention to external and internal performance measures that are...
-
4kg . B 6kg A simple Arwood's machine is shown in the diagram above. It is composed of a frictionless lightweight pulley with two cubes connected by a light string. If cube A has a mass of 4.0 kg and...
-
An object with a mass of 0 , 2 kg rests on a frictionless air table due to a spring force constant k = 5 N / m we pull the mass so that it extends the spring by 0 , 1 m and release it freely at the...
-
(a) Figure 1 shows two points A and B with a straight line drawn through them. A 6 -5 -4 -3 -2 -1 Y 6 5 4 3 2 whe 0 -1 -2 -3 -4 -5 Lov 2 3 4 5 B Figure 1 (i) Write down the coordinates of the points...
-
X X X X R m COM Let'R' be the radius of the cylindrical region in which the magnetic field is assumed to exist. "E' is the induced magnetic field due to the increasing magnetic field B and the rate...
-
Two blocks are connected by a light string that passes over a frictionless pulley. The block of mass m 1 1 = 4 = 4 kg lies on a horizontal surface and is connected to a spring of force constant k =...
-
George has been assigned to be the new project manager for our project. He is eager to get off to a good start and wants to identify what activities he should first engage in. How would you advise...
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
If you look at the history of government grants and subsidies, a large portion go to corporations (over proprietorships and partnerships) even though corporations account for less than 20 percent of...
-
Answer Problem 30.18 for the thermal and photo chemical Cyclizations of (2E, 4Z, 6Z, 8Z)-2, 4, 6, 8-decatetraene.
-
The cyclohcxadccaoctacne shown isomerizes to two different isomers, depending on reaction conditions. Explain the observed results, and indicate whether each reaction is conrotatory ordisrotatory....
-
Which of the following reactions is more likely to occur?Explain. eat eat 2.
-
Provide the codification references to support your answers Scope Questions 1. Which entities are subject to the Interim Reporting guidance within the Codification? 2. Does the Foreign Currency...
-
Why should a company establish an internal control structure? (2 marks) Evaluate the advantages and disadvantages of Perpetual Inventory System against Periodical Inventory System respectively. (2...
-
The Green Chef is an exclusive restaurant in your city. Reservations are difficult to secure and the menu is expensive. The owner, Chef Jorge, is rumored to be running an illegal gambling operation...

Study smarter with the SolutionInn App


