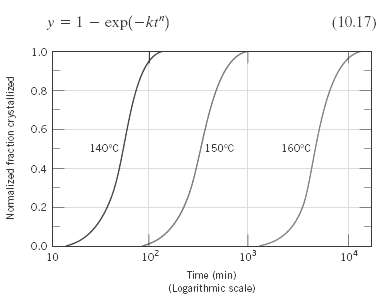
Determine values for the constants n and k (Equation 10.17) for the crystallization of polypropylene (Figure) at
Question:
Determine values for the constants n and k (Equation 10.17) for the crystallization of polypropylene (Figure) at 160?C.
Transcribed Image Text:
y = 1 - exp(-kt") (10.17) 1.0 0.8 0.6 140°C 150°C 160°C 0.4 0.2 0.0 10 10 10? 104 Time (min) (Logarithmic scale) Nomalized frac tion crystallized
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
In this problem we are asked to determine the values of the constants n and k Equation 1017 for the ...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The three bulbs are identical and the two batteries are identical. Compare the brightnesses of the bulbs. A. A>B> C B. A>C> B 000 C. A> B = C D. A < B = C E. A = B = C
-
Determine the constants c and n in Equation 12-2 that describe the rate of crystallization of polypropylene at 140 C. (See Figure 12-30.)
-
Determine the constants c and n in the Avrami relationship (Equation 12-2) for the transformation of austenite to pearlite for a 1050 steel. Assume that the material has been subjected to an...
-
What has motivated you most since accepting your Omega Nu Lambda nomination to grow in your career development? Give an example
-
Write an essay to discuss how compensation programs are developed and how laws can affect compensation practices. Includes the following elements: Issues that influence an individual's decision to...
-
Laubitz Company begins operations on April 1. Information from job cost sheets shows the following: Each job was sold for 25% above its cost in the month following completion. Instructions (a)...
-
A six-cylinder, 4-stroke SI engine delivers \(400 \mathrm{~kW}\) at \(2200 \mathrm{rpm}\). Determine the bore and stroke from the following data : Compression ratio \(=7.6 ;\) stroke to bore ratio...
-
An examination of the cash activities during the year shows the following. GREAT ADVENTURES Cash Account Records July 1, 2024, to December 31, 2024 Date 7/1 7/15 7/22 7/30 8/1 8/10 8/17 9/21 10/17...
-
6) Find the derivative and simplify your answer: e2x y = x +9
-
The Town of Weston has a Water Utility Fund with the following trial balance as of July 1, 2019, the first day of the fiscal year: During the year ended June 30, 2020, the following transactions and...
-
Demonstrate, in a manner similar to Equation 15.4, how vulcanization may occur in a butadienerubber. H; CH; H ---- -C-C=C-C- + (m + n) S- (S)m (S) (15.4) -C-C=C-C- -C-C-C-C- ; H; H
-
Name the following polymer(s) that would be suitable for the fabrication of cups to contain hot coffee: polyethylene, polypropylene, poly (vinyl chloride), PET polyester, and polycarbonate. Why?
-
In Problems 7 26, graph the plane curve whose parametric equations are given, and show its orientation. Find the rectangular equation of each curve. x(t) = t + 4, y(t) = t - 4; t 0
-
From the following information, draw up the trading account section of the income statement of J. Bell for the year ending 31 December 2012, which was his first year in business: Carriage inwards...
-
A On 1 January 2011 a business purchased a laser printer costing 1,800. The printer has an estimated life of four years after which it will have no residual value. It is expected that the output from...
-
Pick a firm such as Bank of America, Patagonia, or L.L. Bean. Develop some potential innovations that would generate a must have. How would you evaluate them?
-
Pick a start-up you admire. What are the major trends emerging from an environmental analysis? What are the major areas of uncertainty? How would a major company in the industry handle these trends...
-
Why didnt Hertz or Avis start an off-airport business directed at insurance companies and vacationers? What advantages would they have had over Enterprise? Why didnt Steinway come up with the...
-
How can attending a job fair help you access the hidden job market?
-
What services are provided by the provincial and territorial governments?
-
Using data from Table 6.5 (page 204), predict the ions that magnesium and aluminum are most likely to form. Table 6.5 Strategy Scan the successive ionization energies for each element to find a point...
-
For the items listed in the following chart, obtain a quantity of 48. Measure the indicated characteristics and determine the process mean and standard deviation. Use a sample size of 4, so that 12...
-
For the data given in Figure 12-3, compute Cp, D, and Cpk, making any assumptions needed to perform the calculations.
-
For the data given in Figure 12-4, compute Cp, D, and Cpk.
-
Compare and contrast different deadlock prevention techniques such as resource ordering, the "hold and wait" condition, and preemptive resource allocation. Which methods are most effective for...
-
The following information is taken from Aden Company's records: Product Group Units Cost/Unit Market/Unit A 1 700 $1.10 $0.90 B 1 250 1.50 1.55 C 2 150 4.90 5.15 D 2 100 6.50 6.40 E 3 80 25.00 24.60...
-
American Food Services, Incorporated leased a packaging machine from Barton and Barton Corporation. Barton and Barton completed construction of the machine on January 1, 2024. The lease agreement for...

Study smarter with the SolutionInn App


