Develop a linear regression equation to predict future demand from the following data: a. Write the regression
Question:
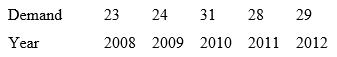
a. Write the regression equation.
b. Predict demand for 2015.
c. Name the independent variable.
d. Name the dependent variable.
Transcribed Image Text:
Demand 23 24 31 28 29 Year 2008 2009 2010 2011 2012
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a b Table for Problem 4 Year Time Period x Actual Demand y x 2 xy 2008 1 23 1 23 2009 2 ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Finance questions
-
Jorge, Jamil, and Jihad are partners in a Limited partnership, Jorge & Jamil are general partners, while Jihad is a limited partner Share capital balances of $50,000, $45,000, and $20,000,...
-
Some researchers3 illustrate a multiple linear regression equation to predict the yearly output of an oil field. We use their latest 20 years of data and change the units of three of their variables...
-
From the following data for liquid nitric acid, determine its heat of vaporization and normal boiling point. Temperature C) Vapor Pressure (mm Hg) 10. 20. 30. 40. 50. 80. 14.4 26.6 47.9 81.3 133 208...
-
Hooghly Products manufactures a silicone paste wax that goes through three processing departments- Cracking, Blending, and Packing. All raw materials are introduced at the start of work in the...
-
Let Y be the rate (calls per hour) at which calls arrive at a switchboard. Let X be the number of calls during a two-hour period. Suppose that the marginal p.d.f. of Y is and that the conditional...
-
Problem refer to the slope field shown in the figure: Show that y = Cx 2 is a solution of dy/dx = (2y)/x for any real number C. 5. -5 -5
-
A printed circuit board has eight different locations in which a component can be placed. If four different components are to be placed on the board, how many different designs are possible?
-
Jessica Company manufactures hockey pucks and soccer balls. For both products, materials are added at the beginning of the production process and conversion costs are incurred evenly. Jessica uses...
-
5. One particular equation involving energy states that E = 1mv where the energy E has 2 units of joules (J), the mass m is in units of kg, and the velocity v is in units of m/s. Another energy...
-
Foster Corporation established Kline Company as a wholly owned subsidiary. Foster reported the following balance sheet amounts immediately before and after it transferred assets and accounts payable...
-
a. Make an exponential smoothing forecast for periods 2 through 5 with 2 values of alpha, 0.05 and 0.60, and an assumed forecast for period 1 of 30. b. Compute the MAD for each of the above...
-
Using the data in table 6-11, calculate a three-month moving average forecast for month 12.
-
Leather Company sold 20,000 units, had variable costs of $12 per unit, fixed costs of $100,000, and profits of $60,000. What is the selling price per unit?
-
3) a) Create a "house" class that initializes house objects in the following way: hl=House(Location,Age,R_Number,Price) Location: Location of the house Age: Age of the house R_Number:Number of the...
-
A petty cash fund was established for $250 dollars on November 1st. On November 30th, the petty cash box had $8.75 remaining and had the following receipts: Postage $98 Office Supplies used during...
-
1. Are the basic accounting principles that all businesses should follow? 2. What is the principle of conservatism in accounting, and how does it impact financial reporting? 3. How does the matching...
-
What are the net benefits of the following teen pregnancy prevention program: The costs of the program were estimated to be $20,000. The benefits were estimated to be $10,000.
-
Share a potential challenge and how your participative/democratic leadership style would be used to solve the issue. Share a challenge or advantage of your participative/democratic leadership style...
-
When the skier of weight W is at point A he has a speed v A . Determine his speed when he reaches point B on the smooth slope. For this distance the slope follows the cosine curve shown. Also, what...
-
When the concentration of a strong acid is not substantially higher than 1.0 10-7 M, the ionization of water must be taken into account in the calculation of the solution's pH. (a) Derive an...
-
Why are new ventures at a disadvantage in receiving debt financing?
-
Why is credit card financing attractive to entrepreneurs? What are the risks?
-
Identify and briefly describe four basic SBA credit programs.
-
Direction: Make an essay about festival dance, chose one from the given questions below. Write your answer in a separate sheet of paper. Processing questions: 1. Why is there a need for festivals?...
-
Stock A has an expected return of 0.1 and a standard deviation of 0.5. Stock B has an expected return of 0.2 and a standard deviation of 0.6. The correlation coefficient is 0.5. 1. What is the...
-
How might the self-fulfilling prophecy work in a positive sense? A negative sense? which is more common, and why? How can negative thoughts like this be realistically managed?

Study smarter with the SolutionInn App


