Develop the feasible product composition regions for the system of figure if the feed composition is 50
Question:
Develop the feasible product composition regions for the system of figure if the feed composition is 50 mol% chloroform, 25 mol% methanol, and 25 mol%acetone.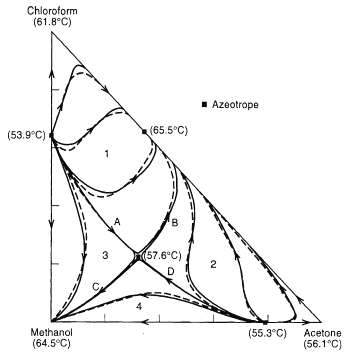
Transcribed Image Text:
Chloroform (61.8°C) 1 Azootrope (65.5°C) (53.9°C) (57.6°C) 3 D. Methanol (55.3°C) Acetone (64.5°C) (56.1°C) B.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The feed composition is shown at F Thus the feed is in Region 1 which is bounded by distillation bou...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Chemical Engineering questions
-
Develop the feasible product-composition regions for the system of figure, using FeedF1. D2 D, (b) B, Region Region 2 (a) B3 B.
-
For the system of figure below find the transfer function G(s)=X 1 (s)/F(s). K1 = 4 N/m K2 = 5 N/m f(t) fy, = 3 N-s/m M =Ikg Jv2=3N-s/m M,= 2 kg fva = 2 N-s/m! %3D
-
For the system of Figure 1, what happens to the two cylinders in each case? a. Push-button 1-PB is momentarily depressed. b. Push-button 2-PB is momentarily depressed. Note that cylinder 2 does not...
-
Oliveras Company had net credit sales during the year of $800,000 and cost of goods sold of $500,000. The balance in accounts receivable at the beginning of the year was $100,000, and the end of the...
-
A $110,000 payment is made on a long-term liability. Of this amount, $10,000 represents interest. Which of the following is not true for the recording of this transaction? a. Reduce liabilities by...
-
People dont want to be managed, they want to be led. Explain what this means and its importance and implications for developing your own style and leadership philosophy.
-
Which correctly represents the flow of information from one financial statement to another? a. Income statement to the statement of retained earnings b. Statement of retained earnings to the balance...
-
A firm wishes to minimize annual inventory costs. The firm uses the EOQ model to determine the cost-minimizing order quantity and the reorder point. Annual demand, units 22,100 Item cost, $ per unit...
-
In July 2 0 2 2 , an individual acquires a rental property for $ 2 0 , 0 0 0 . $ 1 1 0 , 0 0 0 is allocated to the land and $ 1 6 0 , 0 0 0 is allocated to the building. The property is rented for $...
-
Three years ago, the Oak Street TCBY bought a frozen yogurt machine for $8,000. A salesman has just suggested to the TCBY manager that she replace the machine with a new, $12,500 machine. The manager...
-
For the same conditions as Exercise 11.5, use a process-simulation program with the UNIFAC equation to calculate a portion of a distillation curve at 1 atm.
-
Repeat Example 11.3, but with ethanol as the solvent.
-
The following items may appear on a ban statement: 1. EFT payment 2. Note collected for company 3. Bank correction of an error from recording a $7,200 deposit as $2,700 4. Service charge using the...
-
7. A 2.0 kg ball is moving at a velocity of 5.0 m/s. It collides with a stationery ball of mass 2 kg. After the collision the first ball A moves off in a direction 30 to the left of its original...
-
Financing, Forecasting, and Planning In 2011, a firm books the following: increase in cash, $0; increase in inventories $24; increase in accounts receivable, $27; increase in accounts payable, $10;...
-
1. Using a constant amplitude of 30 cm, and a constant length of 1.0 m, measure the time for 10 cycles for masses of 100 g, 500 g and 1000 g. 2. Calculate the period, T and the frequency, f for each...
-
The diagram below shows an oblique force acting on a beam supported by a roller and pin joint: F 30 A 2 m B 1 m If F = 480 N, what is the magnitude of the vertical reaction force at point C in...
-
1 Which of the following statements must be true of the circuit above? A. The voltage across each capacitor is the same. B. The charge on each capacitor must be the same. C. The voltage across C3...
-
Define the following terms: (i) Absolute pressure and Atmospheric pressure. (ii) Enthalpy and Energy.
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
List three factors that can cause a financial crisis.
-
Fifty thousand lb/h of a 20 wt% aqueous solution of NaOH at 120F is to be fed to an evaporator operating at 3.7 psia, where the solution is concentrated to 40 wt% NaOH. The heating medium is...
-
A desublimation unit of the heat-exchanger type is to be sized for the recovery of 200 kg/h of benzoic acid (BA) from a gas stream containing 0.8 mol% BA and 99.2 mol% N 2 . The gas enters the unit...
-
A bar of 98 wt% Al with 2 wt% of Fe impurity is subjected to one pass of zone refining. The solidliquid distribution coefficient for the impurity is 0.29. If z/l = 10 and the resulting bar is cut off...
-
Jeff Getler sold (short) 30 ounces of gold at $1,500/oz and bought (long) 3,000 ounces of silver at $16/oz. The initial and maintenance margins for each position are, $4,500/$2,900 (gold) and...
-
Lauren Wang invented a simple stock index which has futures contracts traded on it. The index is composed of three stocks whose current values are $50, $120 and $80, respectively. The index is...
-
Using IBM balance sheet and income statement, create common-size, common base-year, and common-size, common base-year financial statements. Part IV: Financial Ratios Using IBM financial statements,...

Study smarter with the SolutionInn App


