Draw Fischer projections for these compounds? CHOH a) H-C-CI CH3 COH b) HC-OH A CH3 c) H
Question:
Draw Fischer projections for these compounds?
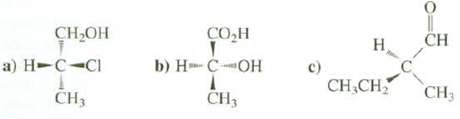
Transcribed Image Text:
CH₂OH a) H-C-CI CH3 CO₂H b) HC-OH A CH3 c) H CH₂CH₂ CH CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
To construct a Fisher projection the molecule is first arranged with the horizontal bonds to ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw Fischer projections for the enantiomers of threo-hexane-1, 2, 3-triol. HOCH2 - CH(OH) - CH(OH) - CH2CH2CH3
-
Draw Fischer projections for the two i aldoheptoses whose stereochemistry at C3, C4, C5, and C6 is the same as that of 1)-glucose at C2, C3, C4, and C5.
-
Draw perspective structures or Fischer projections for the substitution products of the following reactions. (a) (b) (c) CH H-Br + NaCN acetone CH,CH CH water/acetone CH CH2CH, CH,CH, EtOH, heat...
-
Consider the following table, which gives the running time T(n) in seconds for a program for various values of the input size n: a. What is the value of T(n) if n = 160007 10240 5120 40960 20480 B C...
-
Greenlife is a health resort that offers a range of programs for health-conscious managers. Managers who participate are provided with a tailored program, consisting of a series of activities. A job...
-
Chips-R-Us is a computer technology corporation that designs hardware and software for use in large businesses. The corporation regularly pays individuals to install programs and give advice to...
-
Water for a Pelton wheel turbine flows from the headwater and through the penstock as shown in Fig. P12.54. The effective friction factor for the penstock, control valves, and the like is 0.032 , and...
-
CommLine Equipment Corporation specializes in the manufacture of communications equipment, a field that has become increasingly competitive. Approximately two years ago, Ben Harrington, president of...
-
Explore and discuss the provisions and requirements of legislation or international standards such as the Payment Card Industry - Data Security Standard (PCI-DSS) Official PCI Security Standards...
-
The Department of Corrections (DOC) needs to replace two of its prison transport buses. There are two companies that have a track record of making reliable vehicles, and both make a bus that meets...
-
Explain whether these compounds rotate plane-polarized light: H a) HC-CCH, HC Br c) Br J. s CI b) Cl CI d) HC -CH3
-
Assign the configurations of the compounds represented by these Fischer projections as R or S. CH -CH2CH3 b) CH2=CH- ) >N- CH-OH CH3 Serine
-
Partnership EFGH is a calendar year, accrual basis partnership. Partner G is a calendar year, cash basis partner. Partnership EFGH rents property from Partner G. EFGH pays market rate rent of $4,000...
-
What is trade discount and what is meant by the successive way of calculating the multiple discount? Give a numerical example to illustrate your answer.
-
List at least four examples of each of the firm's cash inflow and its cash outflow.
-
What is debt capital and equity capital, and how would a firm decide to depend on any of them?
-
What is account receivable aging and what purpose would it serve?
-
What are the major harvesting strategies? Briefly explain each and every one of them.
-
In this chapter, we derived the canonical commutation relation between the position and momentum operators, but we had previously constructed the derivative operator \(\mathbb{D}\) on a grid of...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Calculate y" and y"'. y = 4t 3 9t 2 + 7
-
Assign R or S configuration to the chirality center in each of the followingmolecules: CH (c) . (b) (a) 2 "- HS efpo -- CH-
-
Draw a tetrahedral representation of (S)-2-pentanol (2-hydroxypentane).
-
Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (blue = N, yellow =S):
-
Could Arthur Murray have created language that would have avoided the application of impossibility of performance? What if Parker was not incapac-itated but that it was simply painful to dance after...
-
In the text's case on the county clerk and marriage licenses, whatare the ethical issues involved? What are the conflicting ethical values and whichvalues were maximized/minimized? The Case of the...
-
2 Altira Corporation provides the following information related to its inventory during the month of August 2024: August 1 Inventory on hand-2,000 units%;B cost $5.30 each. August 8 Purchased 8,000...

Study smarter with the SolutionInn App


