Electrophilic addition of Br2 to isoprene (2-methyl-1, 3-hutadiene) yields the following product mixture: Of the 1, 2-addition
Question:
Electrophilic addition of Br2 to isoprene (2-methyl-1, 3-hutadiene) yields the following product mixture: Of the 1, 2-addition products, explain why 3, 4-dibromo-3-methyl-1-butene (21 %) predominates over 3, 4-dibromo-2-mcthyl-1-butene(3%).
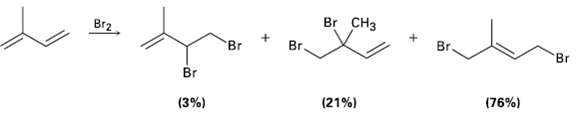
Transcribed Image Text:
Br CHз Br2 Br. Br Br 'Br Br (76%) (21%) (3%)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (9 reviews)
CH3 8 8 BrCH CCHCH A tertiaryprimary allylic carboca...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The rate law for addition of Br2 to an alkene is first order in Br2 and first order in the alkene. Does this fact prove that the mechanism of addition of Br2 to an alkene proceeds in the same manner...
-
Explain why methyl propyl ether forms both methyl iodide and propyl iodide when it is heated with excess HI.
-
Explain why the following reaction yields the Hofmann product exclusively (no Zaitsev product at all) even though the base is not sterically hindered: Br NaOEt ELOH
-
Recall the heat equation which we solved numerically T= = DTxx There we implemented an explicit numerical scheme (FTCS) which led to a conditionally stable solution - meaning that for certain time...
-
Would an employee who posts, "I hate my job!" be protected, according to the principles cited here? Why or why not?
-
(a) Will Co(OH)2 precipitate from solution if the pH of a 0.020 M solution of Co(NO3)2 is adjusted to 8.5? (b) Will AgIO3 precipitate when 20 mL of 0.010 M AgNO3 is mixed with 10 mL of 0.015 M NaIO3?...
-
An auditor is performing an attribute estimation sampling plan. The risk of assessing control risk too low is .05 and sample size is 80. Assuming one deviation is detected, what is the auditor's...
-
Flight Caf is a company that prepares in-flight meals for airlines in its kitchen located next to the local airport. The companys planning budget for July appears below: In July, 17,800 meals were...
-
What are the the differences between a Micro-manager and an Investor. Provide examples of each.
-
1. Develop a simulation model for 52 weeks of operation at Ebony. Graph the inventory of soap over time. What is the total cost (inventory cost plus production change cost) for the 52 weeks? 2. Run...
-
Treatment of 3, 4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how...
-
Propose a structure for a conjugated diene that gives the same product from both 1, 2- and 1, 4-addition of HBr.
-
Write down the first five terms of the sequence. {()'} {s} (3,
-
1. In any case, a plaintiff must establish standing to sue. That means they must have suffered an actual harm and the harm must be redressable. Think about why this standing requirement exists....
-
Let u = (2,1,1,2), a = (4,-4,2,-2). Find projau and u-projau.
-
Explain how law, policy, and regulation influence the way that people and organizations communicate.
-
Keeping in mind Endo's views of Jesus and the nature of Japanese people, evaluate the effectiveness of Endo's short fiction in presenting his spiritual views. Discuss the four (4) stories we read in...
-
How do companies like TikTok create change? How did Google create change when it was first developed? What do the changes that these companies have brought to the world show about how technology has...
-
If each major grid unit in Figure 10.9 corresponds to \(1 \mathrm{~m}\), specify the location of point \(\mathrm{P}\) in terms of its \(x\) and \(y\) coordinates in each of the three coordinate...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
The enthalpy of solution for NaOH is -44.46 kJ/mol. What can you conclude about the relative magnitudes of the absolute values of Hsolute and Hhydration , where Hsolute is the heat associated with...
-
Predict the major products of the following reactions. (a) 2, 4-dinitrochlorobenzene + NaOCH3 (b) Phenol + tert-butyl chloride + AlCl3 (c) Nitrobenzene + fuming sulfuric acid (d) Nitrobenzene +...
-
Predict the major products of bromination of the following compounds, using Br2 and FeBr3 in the dark. (a) (b) (c) NO, OCH OCH3 OCH,
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
The following information pertains to Mason Company for Year 2 . Beginning inventory 1 1 6 units @ $ 3 0 Units purchased 3 5 8 units @ $ 4 5 Ending inventory consisted of 4 2 units. Mason sold 4 3 2...
-
Luzadis Company makes furniture using the latest automated technology. The company uses a job - order costing system and applies manufacturing overhead cost to products on the basis of machine -...
-
Thoroughly define organizational culture and provide three examples of different types of organizational cultures. Several sources and examples support thinking. explain how leaders and managers...

Study smarter with the SolutionInn App


