This elimination reaction gives a single product. Show its structure and explain why it is the only
Question:
This elimination reaction gives a single product. Show its structure and explain why it is the only productformed.
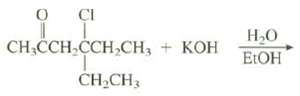
Transcribed Image Text:
O ÇI CH,CCH CCH,CH, + KOH Н-О кон ELOH ČH,CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
Because of the relatively acidic hydrogen on ...View the full answer

Answered By

Rishabh Ojha
During my undergraduate i used to participate as TA (Teaching Assistant) in several electronics and computers subject. I'm passionate about learning Computer Science as my bachelors are in Electronics but i learnt most of the Computer Science subjects on my own which Machine Learning also. At Present, i'm a working professional pursuing my career as a Machine Learning Engineer and i want to help others learn during my free hours, that's all the motivation behind giving tuition. To be frank i have no prior experience of tutoring but i have solved problems on opensource platforms like StackOverflow and github. ~Thanks
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The reaction shown gives a single product in 88% yield. What is that product? OCH3 AICI CH3O (CH3)2CHCH2CCl OCH3
-
The reaction shown gives a single product in 88% yield. What is that product?
-
Explain why it is important for all departments of an organizationmarketing, accounting, finance, operations management, human resources, and so onto think consumer. Why is it important that even...
-
The common stock of Scarlet Enterprises currently pays a quarterly dividend of $1.75 per share. The dividend is assumed to grow annually at a constant rate of 4.0%. The market beta for Scarlet...
-
Garden State Markets, a chain of convenience grocery stores, has store hours that fluctuate from month to month as the tourist trade varies. The electricity costs and hours of operation for one of...
-
The Information Systems Audit and Control Association surveyed office workers to learn about the anticipated usage of office computers for personal holiday shopping (USA today, November 11, 2009)....
-
Suppose Ironman, Inc., lost all of its inventory in a flood. Beginning inventory was \($43,000,\) net purchases totaled \($524,000,\) and sales came to \($875,000.\) Ironmans normal gross profit...
-
Alina Sadofsky, who operates a small grocery store, has established the following policies with regard to the checkout cashiers: 1. Each cashier has his or her own cash drawer, to which no one else...
-
Wyckam Manufacturing Incorporated has provided the following estimates concerning its manufacturing costs: Fixed Cost per Month Cost per Machine-Hour Direct materials $ 5.50 Direct labor $ 42,800...
-
The potential of employing two or more helicopters for transporting payloads that are too heavy for a single helicopter is a well-addressed issue in the civil and military rotorcraft design arenas...
-
When 2-bromo-2-methylbutane is heated in a mixture of ethanol and water, it gives a 64% yield of substitution products and a 36% yield of elimination products. (a) What mechanisms does this reaction...
-
How might the structure of DDT be modified to make it again effective against resistant insects?
-
Establish the mechanical energy balance equation starting from the law of conservation of energy for a control volume.
-
We have a non-rotating space station in the shape of a long thin uniform rod of mass 4.45 x 10^6 kg and length 1387 meters. Small probes of mass 9598 kg are periodically launched in pairs from two...
-
A diver of mass 63.3 kg stands on one end of a diving board of mass 35.9 kg, as shown in the diagram. Everything is in equilibrium (since nothing is moving). What is the magnitude of the support...
-
Requirements Calculate the net undiscounted and discount cash flows that you would use in the impairment tests. Explain why the discount rate used to calculate discounted cash flows is appropriate...
-
Tom and John go to an island to fish and then bring to the market to sell. They are the only people to sell finish in the market which has the following demand: P = 100 - Q, where P is the price and...
-
An investor just purchased a semi-annual coupon bond with a coupon rate of 5% and $1,000 face value. The bond has a maturity is 20 years, and its yield to maturity is 8 percent. a) Find the...
-
In the rough sea of Chapter Problem 4.47, what is the required stiffness of an elastic foundation of damping ratio 0.05 to limit the acceleration amplitude of a \(5-\mathrm{kg}\) radio set to \(1.5...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
Find the slope of each line. y x
-
Allene, H2C = C = CH2, has a heat of hydrogenation of 298kJ/mol (71.3 kcal/mol). Rank a conjugated diene, a non-conjugated diene, and an allene in order of stability.
-
Give the structures of both 1, 2 and 1, 4 adducts resulting from reaction of 1 equivalent of HC1 with 1, 3-pentadiene.
-
Look at the possible carbocation intermediates produced during addition of HCI to 1, 3-pentadiene (Problem 14.2), and predict which 1, 2 adduct predominates. Which 1, 4 adduct predominates?
-
Flounder Company reported the following information for 2019: October November December Budgeted sales $300,000 $334,000 $378,000 Budgeted purchases $120,000 $127,000 $144,500 All sales are on...
-
Concord Co. decides at the beginning of 2025 to adopt the FIFO method of inventory valuation. Concord had used the LIFO method for financial reporting since its inception on January 1, 2023, and had...
-
An experimental rocket sled accelerates from 12.0 m/s to 280 m/s in 4.70 seconds. How far did it travel in this time?

Study smarter with the SolutionInn App


