Ethers can often be prepared by S N 2 reaction of alkoxide ions, RO ? , with
Question:
Ethers can often be prepared by SN2 reaction of alkoxide ions, RO?, with alkyl halides. Suppose you wanted to prepare cyclohexyl methyl ether. Which of the two possible routes shown below would you choose? Explain.
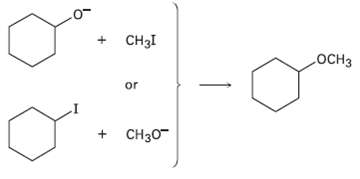
Transcribed Image Text:
CH3I .ОCHЗ or + CH30
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
CH3I OCH3 CH30 This is an excellent method of ether preparat...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Suppose you wanted to test H0: p = 0.50, but you had 0 successes in n trials. If you had found the test statistic using the se = p(1 - p)/n designed for confidence intervals, show what happens to the...
-
Suppose you wanted to know if men or women students spend more money on clothes. You consider two different plans for carrying out an observational study: Plan 1: Ask the participants how much they...
-
Suppose you wanted to test your extrasensory perception (ESP) ability using an ordinary deck of 52 cards, which has 26 red and 26 black cards. You have a friend shuffle the deck and draw cards at...
-
Identify how balance sheet quality and earnings quality were impaired in each of the following accounting scandals: Waste Management 1988 - Falsely increased the useful lives of long-lived tangible...
-
Keeping in mind that an agency like LTSC would have limited funding and just a few senior managers, suggest two additional developmental activities that are likely to be most beneficial to the...
-
What is the time and space complexity of fuzzy c-means? Of SOM? How do these complexities compare to those of K-means?
-
A chi square test is easily implemented on a computer. With the counts from Example 8 in columns 1-4, the MINITAB commands produce the output Expected counts are printed below observed counts Repeat...
-
At the end of its first year, the trial balance of Bronowski Company shows Equipment $30,000 and zero balances in Accumulated DepreciationEquipment and Depreciation Expense. Depreciation for the year...
-
You have a staff of three (3) uniformed personnel in your office that can conduct inspections and fire prevention activities. Additionally you have eleven (11) uniformed staff on duty that can...
-
Refer to the Lincolnville School District bus data. First, add a variable to change the type of engine (diesel or gasoline) to a qualitative variable. If the engine type is diesel, then set the...
-
Reaction of the following S tosylate with cyanide ion yields a nitrile product that also has S stereochemistry.Explain. TOS NaCN |" CH2OCH3 (S stereochemistry)
-
We saw in Section 7.8 that bromohydrin are converted into epoxides when treated with base. Propose a mechanism, using curved arrows to show the electronflow. Br -C CH3 CH NAOH Ethanol TH CH C
-
Point for Discussion: Because Bob had not been served or made aware of the order, should he be held accountable for violating the order by withdrawing the funds?
-
The risk free rate is currently 2% and the required return on the market portfolio is 8%.If a specific stock is estimated to have a CAPM beta coefficient of 1.2 and the $2.00 per share dividend paid...
-
Your client, Quyen, is 25 years old, single and a non-smoker. She has just received a quote from an insurance company for $1 million in term life insurance. The annual premium would be $1,345....
-
Eduwize Ltd is a comprehensive, listed and accredited education provider that owns a variety of educational institutions from preschools to universities, offering qualifications up to master's degree...
-
China is an emerging market. Research if investing in that market is a good or bad investment right now. Provide supporting articles or evidence supporting this position. What is the future forecast...
-
You are evaluating an investment with a purchase price (outflow) of $100 today. However, you expect a $120 inflow at the end of each of the next 2 years. Assuming 5% nominal rate, compounded...
-
What is the effective annual interest rate if the nominal annual interest rate is 24 percent per year compounded monthly? a. 2 percent b. 24 percent c. 26.82 percent d. 27.12 percent
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
Consider the reaction: An equilibrium mixture of this reaction at a certain temperature has [NH 3 ] = 0.278 M and [H 2 S] = 0.355 M. What is the value of the equilibrium constant (K c ) at this...
-
(a) Propose a mechanism for the reaction of benzyl alcohol with acetyl chloride to give benzyl acetate. (b) Propose a mechanism for the reaction of benzoic acid with acetyl chloride to give acetic...
-
The mass spectra of acid derivatives follow the principles shown in Chapter 18 for other carbonyl compounds and for alkoxy groups. Both McLafferty rearrangements and alpha-cleavages are common. The...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
Supply is the connection between the cost of a decent and the amount ready to move costing that much. It very well might be addressed as a table or diagram relating cost and amount provided. Makers,...
-
If 50% of the men 18-34 years old watch television on Mondays at 11 PM, and 30% of those viewers watch Monday Night Football, what is Monday Night Football's rating among men 18-34?
-
Assume you are a media planner and you have just completed a media plan using both magazine and television advertising. Your magazine schedule reaches 50 percent of the target audience and your...

Study smarter with the SolutionInn App


