Evidence for restricted rotation around amide CO?N bonds comes from NMR studies. At room temperature, the 1
Question:
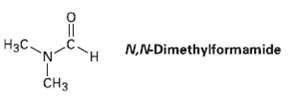
Evidence for restricted rotation around amide CO?N bonds comes from NMR studies. At room temperature, the 1H NMR spectrum of N, N-dimethyl form amide shows three peaks: 2.9 ? (singlet, 3 H), 3.0 ? (singlet, 3 H), 8.0 ? (singlet, 1 H). As the temperature is raised, however, the two singlets at 2.9 ? and 3.0 ? slowly merge. At 180 ?C, the 1H NMR spectrum shows only two peaks: 2.95 ? (singlet, 6 H) and 8.0 ? (singlet, 1 H). Explain this temperature-dependent behavior.
Transcribed Image Text:
N,N-Dimethylformamide Нзс. CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
H NMR shows that the two methyl groups of NNdimethylformamide are nonequ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum of compound A (C8H8O) consists of two singlets of equal area at 5.1 (sharp) and 7.2 ppm (broad). On treatment with excess hydrogen bromide, compound A is converted to a single...
-
The 1H NMR spectrum of N,N-dimethylformamide shows a separate signal for each of the two methyl groups. Can you explain why?
-
The 1H NMR spectrum of the product obtained when an unknown alkene reacts with ozone and the ozonolysis product is worked up under oxidizing conditions is shown. Identify the alkene. 10 (ppm)...
-
Suppose a consumer lives in two periods , with his income in period 1 as $100 and his income in period 2 as $150. If the rate of interest in the economy is 12%. Find the equilibrium level of...
-
In what three situations should you consider using passive voice?
-
Compare the levels of dynamic impedance for the 24-V diode of Fig. 1.48b at current levels of 0.2, 1, and 10 mA. How do the results relate to the shape of the characteristics in this region?
-
For the revised propane refrigeration cycle in Figure 10.21, let the isentropic efficiencies of the turbine and compressor be 0.9 and 0.7 , respectively. Compute the following: (a) Lost work for the...
-
The ultimate analysis of a No.4 fuel oil is 86.47 wt% carbons. 11.65% hydrogen, 1.35% sulfur, and the balance noncombustible inerts this oil is burned in a steam-generating furnace with 15% excess...
-
Discuss/debate with your classmates about the early history of the criminal theory and its application to crime control i.e., Positivist School vs. Classical School, plus gender, class, and race as...
-
Refer to the data set pit.csv. Find a confidence interval of the specified level for the mean depth of pits for the following durations and humidity levels. When the sample size is 30 or less,...
-
Cytochrome c is an enzyme found in the cells of all aerobic organisms. Elemental analysis of cytochrorne c shows that it contains 0.43% iron. What is the minimum molecular weight of this enzyme?
-
Propose a structure for an octapeptide that shows the composition Asp, Gly2 Leu, Phe, Pro2, Val on amino acid analysis. Edman analysis shows a glycine N-terminal group, and leucine is the C-terminal...
-
Explain very briefly the function of following mountings: (i) Steam stop valve (ii) Feed check valve (iii) Blow-off cock (iv) water level indicator (v) Pressure gauge (vi) Safety valve
-
provide a brief financial summary about the company's Wicked Good Cupcakes and American Airlines. (Links will be provided for both companies, you may have to copy and paste into the browser to view...
-
Question 5 (5 points) If $25,000 is invested in a savings account offering 3.25% per annum, compounded monthly. How fast is the balance growing, to the nearest dollar, two years later?
-
9. Construct a quantum circuit to add two two-bit numbers x and y modulo 4. That is, the circuit should do |x,y)|x,x+y mod 4). 1
-
Assumptions Historicals Stage 1 Stage 2 Assumption 2017 2018 2019 Average 2020 2021 2022 2023 2024 2025 2026 2027 2028 2029 Revenue Growth XYZ Tech ...
-
2) Show that ||xx2x|1 for all x R^, and that equalities can occur, even for nonzero vectors.
-
How much money must be deposited now in order to withdraw $10,000 in 4 years if interest is 5 percent compounded quarterly?
-
Factor and simplify, if possible. Check your result using a graphing calculator. 3 cot 2 + 6 cot + 3
-
Caffeine (C 8 H 10 N 4 O 2 ) is a weak base with a pKb of 10.4. Calculate the pH of a solution containing a caffeine concentration of 455 mg/L.
-
Give a synthesis of from aniline. NHCH2CHs
-
Give a synthesis for from toluene. H2N CH3 NH2
-
Show how CH3CH2N(CH3)2 can be synthesized from an amide.
-
On January 1 of Year 1, Innovative Lab issued a 4-year $50,000 note to a local bank with fixed interest payments based on 6%, payable annually on December 31. To hedge the risk of a fixed interest...
-
Part 4 Tennis company, a distributor of Tennis Rackets, has been experiencing losses for some time, as shown by its most recent monthly contribution format income statement below: Sales Variable...
-
Bunnell Corporation is a manufacturer that uses job-order costing. On January 1, the company's inventory balances were as follows: Raw materials Work in process Finished goods $ 59,500 $ 37,600...

Study smarter with the SolutionInn App


