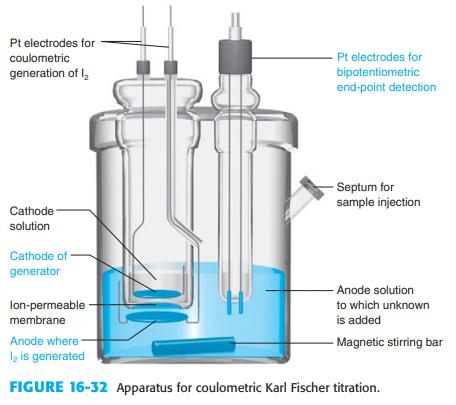
Explain how the end point is detected in a Karl Fischer titration in Figure 16-32. Figure 16-32
Question:
Explain how the end point is detected in a Karl Fischer titration in Figure 16-32.
Figure 16-32
Transcribed Image Text:
Pt electrodes for coulometric Pt electrodes for generation of l, bipotentiometric end-point detection - Septum for sample injection Cathode solution Cathode of generator - Anode solution lon-permeable - to which unknown membrane is added Anode where l2 is generated - Magnetic stirring bar FIGURE 16-32 Apparatus for coulometric Karl Fischer titration.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
The bipotentiometric detector maintains a constant current ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Write the chemical reactions that show that 1 mol of I 2 is required for 1 mol of H 2 O in a Karl Fischer titration.
-
Explain how the amperometric end-point detector in Figure 16-8 operates. Figure 16-8 To constant-current coulometric power supply Port for adding reagents, with ground-glass stopper Microammeter Pt...
-
Explain how a molecule is assigned to a point group.
-
Write a recursive program to draw plasma clouds, using the method suggested in the text.
-
Lofton Company purchased a delivery truck. The total cash payment was $27,900, including the following items. Negotiated purchase price .......... $24,000 Installation of special shelving ..........
-
Debt to Equity = total debt / total equity Year 2 = 316,632/146,043 Year 1 = 282,304/138,245 = 2.17 = 2.04 1. What does this ratio tell you? 2. In general, would you like this ratio to be higher or...
-
The phase-plane equation of a single-degree-of-freedom system is given by \[\frac{d y}{d x}=\frac{-c y-\left(x-0.1 x^{3} ight)}{y}\] Investigate the nature of singularity at \((x, y)=(0,0)\) for...
-
The following information concerns production in the Baking Department for July. All direct materials are placed in process at the beginning of production. a. Determine the number of units in work in...
-
Consider the following financial statement information for the Newk Corporation: Item Inventory Accounts receivable Accounts payable Credit sales Beginning $ 11,000 6,000 Ending $ 12,000 6,300 8,200...
-
Students measured the concentration of HCl in a solution by titrating with different indicators to find the end point. Is the difference between indicators 1 and 2 significant at the 95% confidence...
-
What is the purpose of the Nafion membrane in Figure 16-30? Figure 16-30 40
-
Consider the following electrolysis reactions. (a) Calculate the voltage needed to drive the net reaction if current is negligible. (b) Suppose that the cell has a resistance of 2.0 and a current of...
-
Apollo Companys sales manager predicts that annual sales of the companys product will soon reach 40,000 units and its price will increase to $200 per unit. According to the production manager, the...
-
There are 5 taxis working at a taxi stop. The mean travel time from leaving the stop to coming back to stop takes 1 hour on average. On the other hand, the mean time between the arrivals of...
-
Question 1: Take a piece of paper and draw 3 graphs: 1. the velocity as function of the time of a car, knowing that: Between 0 and 2 seconds, the velocity of the car is v (t) = 20 x t 3 Between 2 and...
-
5. A researcher is investigating the realtionship between US GDP and Consumption growth. The data, which has been collected quarterly and is from 1960 to 2019, is plotted below: GR US CU and...
-
Vector A has magnitude 5.00 and is at an angle of 36.9 south of east. Vector B has magnitude 6.40 and is at an angle of 20.0 west of north. Choose the positive x-direction to the east and the...
-
PROBLEM-3 You have set Cruise Control set to 10 m/s (traveling at constant velocity). Assume that the road you are driving on has negligible friction and that you can ignore the effect of the rain...
-
Mr. Tamm has $9000 to invest in three types of stocks: low-risk, medium-risk, and high-risk. He invests according to three principles. The amount invested in low-risk stocks will be at most $1000...
-
Write a paper about medication error system 2016.
-
Using activities, calculate the pH and concentration of H + in 0.050 M LiBr at 25 C.
-
Using activities, calculate the pH and concentration of H + in 0.050 M LiBr at 25C.
-
A 40.0-mL solution of 0.040 0 M Hg 2 (NO 3 ) 2 was titrated with 60.0 mL of 0.100 M KI to precipitate Hg 2 I 2 (K sp = 4.6 10 -29 ). (a) Show that 32.0 mL of KI are needed to reach the equivalence...
-
You are planning for retirement in 20 years and currently have $85,000 in a savings account and $25,000 in a mutual fund. Assuming your savings account returns 4% compounded semi-annually, while your...
-
An Italian company is expecting to receive Kuwait dinars in 9 months' time. The spot rate is Lire/dinar 5.467.Your Company could borrow in dinar at 9% per annum or in Lire at 14% per Annum No forward...
-
Gold coast organics has a total assets turnover of 2.5 and a net profit margin of 3.5 percent.the total debt ratio for the firm is 50 percent.calculate the gold coast organics return On equity.

Study smarter with the SolutionInn App


