Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
Question:
Explain whether the methyl is axial or equatorial in this compound:
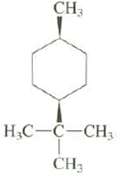
Transcribed Image Text:
CH3 H C-C-CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The axial destabilization energy of the ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
The compound below is an example of a methyl ester. Methyl esters react with lithium iodide to give lithium carboxylate salts. The solvent in this example is pyridine (margin). Suggest several...
-
Explain why each compound or ion should be aromatic, antiaromatic, or nonaromatic. (a) The cyclonona-tetraene cation (b) The cyclononatetraene anion (c) The [16]annulene dianion (d) The [18]annulene...
-
The firm is considering between two mutually exclusive projects, Project A and Project B, that each require an upfront investment of $100m and are expected to produce free cash flow (FCF) only at the...
-
Predetermined plantwide overhead rate; alternative cost drivers Repeat the requirements for Exercise 7.22, assuming that Facsimile uses the following as overhead cost drivers: (a) Direct labour...
-
Compute net cash flow from operating activities for Tulsa Corporation under the indirect reporting format. Tulsa Corporation provided you with the following information for the current year. Compute...
-
What are the key components of the Equipment Element? Give examples.
-
Stallman Company took a physical inventory on December 31 and determined that goods costing $200,000 were on hand. Not included in the physical count were $25,000 of goods purchased from Pelzer...
-
Metlock Tackle Co. Ltd. reports the following inventory transactions for its fishing rods for the month of April. The company uses a perpetual inventory system. Date Explanation Units Unit Cost/Price...
-
Rose-comb chickens mated with walnut-comb chickens produced 15 walnut-, 14 rose-, 5 pea-, and 6 single-comb chicks. Determine the genotypes of the parents.
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is...
-
Explain the wave behavior known as diffraction. Draw the diffraction pattern that occurs when light travels through two slits comparable in size and separation to the lights wavelength.
-
For the vectors shown below where v V2 3, suppose you wanted to determine the values of A and B that would make Av Bv = -23. What are the two equations you would use? You should use, for example,...
-
Create one personal financial plan for three exemplar couples. Any gender is allowed. Couple 1: Aged 28 &32 With 2 Children under age of 5. Their combined net income is $55,000 p/a, total expenditure...
-
Q #1 (Corporate income tax) The William B. Waugh Corporation is a regional Toyota dealer. The firm sells new and used trucks and is actively involved in the parts business. During the most recent...
-
Why does the call: System.out.println(System.out): java.io.Printstream@7a84e4? produce a result such as 1) Because the implementor of the PrintStream class supplied a toString method 2) Because the...
-
Add the following numbers in the order given to hash table size 7. 16-12-25-15-2 The function is %5; the second function is +3. A. B. 25 O E. 2 0 2 0 16 0 1 25 16 12 O C. The correct hash table is...
-
What are the two primary discounted cash flow (DCF) methods for project evaluation?
-
Could a set of three vectors in span all of? Explain. What about n vectors in when n is less than m? R4
-
Find (/g)'(1) if f (1) = '(1) = g(1) = 2 and g'(1) = 4.
-
Qiana, a polyamide fiber with a silky texture, has the following structure. What is the monomer units used in the synthesis ofQiana? flomdoCor CH2- Qiana CICH2)6C-NH- NH-
-
What is the structure of the polymer produced by treatment of ?-propiolactone with a small amount of hydroxide ion? B-Propiolactone
-
Polyimides having the structure shown arc used as coatings on glass and plastics to improve scratch resistance how would you synthesize a polyimide? A polyimide -N-
-
es Hart, Attorney at Law, experienced the following transactions in Year 1, the first year of operations: 1. Accepted $16,600 on April 1, Year 1, as a retainer for services to be performed evenly...
-
Dahlia Corporation has a current accounts receivable balance of $439,516. Credit sales for the year just ended were $5,503,810. a. What is the receivables turnover? Note: Do not round intermediate...
-
Why does the organizational structure hold political significance? Provide an in-depth analysis of this concept using examples from both academic literature and real-world instances. Additionally,...

Study smarter with the SolutionInn App


