Explain which compound has a faster rate of SN1 reaction. a) c) HC CI CI or or
Question:
Explain which compound has a faster rate of SN1 reaction.
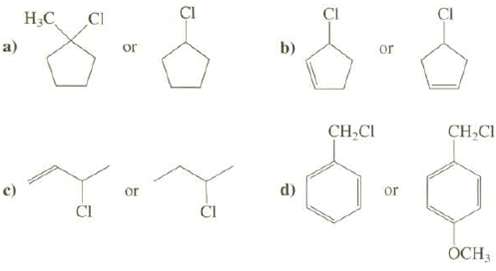
Transcribed Image Text:
a) c) H₂C CI CI or or J d) CI CH₂CI or or D CH₂Cl OCH 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
For the S N 1 reaction formation of the carbocation is the rate limitin...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which compound has a faster rate of reaction withHCI: b) or or NO2 or
-
Explain which compound has a faster rate of SN2 reaction: a) HC CI CI c) PhCHCH3 or H CI Cl or CHCHCH3 b) CI CH3 CH3 or U
-
Which of these compounds would have faster rate of SN1 reaction? a) b) c) d) e) CI ta Ph Ph -CI or Br or Br or or -Cl or + CH3 Ph -Cl Br Br ta -CI
-
What other types of contingency planning should Matt and Chris include to make the report comprehensive? Please explain the relevance of each suggestion.
-
Describe the problems that can arise in using process costing to estimate the cost of the services produced in mass service entities.
-
Carver Corporation uses the accrual method of accounting and the calendar year as its tax year. Its board of directors authorizes a cash contribution on November 3 of Year 1, that the corporation...
-
Water is pumped between the two tanks described in Example 12.4 once a day, 365 days a year, with each pumping period lasting two hours. The water levels in the two tanks remain essentially constant....
-
New Centurys bankruptcy examiner charged that KPMG did not comply with applicable professional standards while auditing the company. List specific auditing standards or principles that you believe...
-
The total sales of a company is $ 1 , 0 0 0 K . The company aims to increase its profit by 2 5 % . There are two ways to increase profit by 2 5 % , either by purchasing the raw materials at a lower...
-
Given IDSS = 9 mA and VP = -3.5 V, determine ID when: a. VGS = 0V. b. VGS = -2V c. VGS = -3.5V d. VGS = -5V
-
Consider the free energy versus reaction progress diagram for the SN2 reaction shown in Figure 8.1. Does the transition state for this reaction have the C Cl bond less than half broke, approximately...
-
Arrange these compounds in order of decreasing SN1 reaction rate. Ph CI CI CI Ph CI Ph
-
VMware ESXi has been made very small. Why? After all, servers at data centers usually have tens of gigabytes of RAM. What difference does a few tens of megabytes more or less make?
-
In an \(L C\) circuit, there is always some energy dissipation as electrical energy is converted to thermal energy. (a) What property of the circuit is responsible for electrical energy being...
-
1. You are a new administrator at Jonestown Medical Center. You receive a telephone call from the nurse manager of the emergency room. Dr. Smith, an emergency room physician who is an employee of...
-
A feed stream containing a mixture of 40% n-butane, 30% n-pentane, and 30% n-hexane fl ows into a fl ash unit. The fl ash temperature is 290 K and the fl ash pressure is 0.6 bar. What is the ratio of...
-
A circuit consists of an AC source, for which \(\mathscr{E}_{\max }=\) \(18 \mathrm{~V}\), in series with a \(1200-\Omega\) resistor and a capacitor. If the amplitude of the potential difference...
-
Darshan's management is pondering whether to lease the machine for \($3,000\) per year or whether to buy it outright. The company MARR is 12%. Problem data is gathered below. What is your...
-
With H 1 : p Assume that the significance level is = 0.05; use the given statement and find the P-value and critical values. P-value = area to the left of the test statistic Left-tailed Left Start...
-
Tanaka Company's cost and production data for two recent months included the following: March April Production (units).........300................600 Rent.....................$1,800............$1,800...
-
Find the slope of the line through each pair of points. (8,9), (8, 16)
-
How would you prepare ds-2-butene starting from propyne, an alkyl halide, and any other reagents needed? This problem cant be worked in a single step. Youll have to carry out more than one reaction.
-
Beginning with 4-octyne as your only source of carbon, and using any inorganic reagents necessary, how would you synthesize the following compounds? (a) cis-4-Octene (b) Butanal (c) 4-Bromooctane (d)...
-
Beginning with acetylene and any alkyl halides needed, how would you synthesize the following compounds? (a) Decane (b) 2, 2-Dimethylhexane (c) Hexanal (d) 2-Heptanone
-
An investor put 60 percent of his money into a risky asset offering a 10 percent return with a standard deviation of return of 8 percent, and he put the balance of his risk-free asset offering 5...
-
There is a bonus with the following characteristics: nominal value $98.650.000 annual coupon rate 4.5% term 5 years, tir 6.5% A) Calculate the price of the bond assuming it is zero coupon B)...
-
Ida Company produces a handcrafted musical Instrument called a gamelan that Is similar to a xylophone. The gamelans are sold for $949. Selected data for the company's operations last year follow:...

Study smarter with the SolutionInn App


