Explain which functional group(s) is present in the compound that has this IR spectrum: 80 60 40
Question:
Explain which functional group(s) is present in the compound that has this IR spectrum:
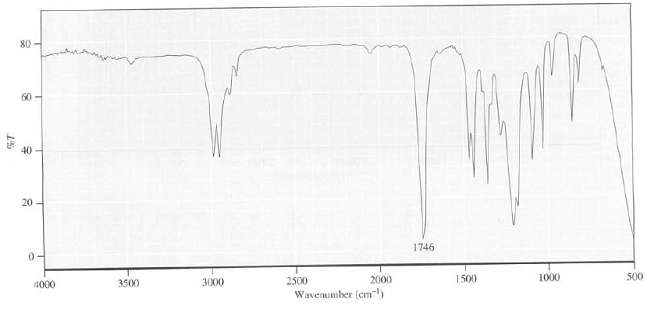
Transcribed Image Text:
80 60 40 20 - 1746 0- 500 1500 1000 2000 2500 3000 3500 4000 Wavenumber (cm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
The bands 30002850 cm 1 region show the p...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Compound A has molecular formula C 8 H 8 O. An IR spectrum of compound A exhibits a signal at 1680 cm -1 . The 1 H NMR spectrum of compound A exhibits a group of signals between 7.5 and 8 ppm (with a...
-
List all types of bonding present in the compound CaCO3. List all types of bonding present in the compound CaCO3. I. ionic bond II. polar covalent bond III. nonpolar covalent bond A. I only B. II...
-
The cesium isotope 137Cs is present in the fallout from aboveground detonations of nuclear bombs. Because it decays with a slow (30.2y) half-life into 137Ba, releasing considerable energy in the...
-
Which of the following is not one of the techniques used in webmining? a. Content mining b. Structure mining c. Server mining d. Usage mining e. Data mining Which of the following would you use to...
-
State corporate income tax laws. Go to the Internet site of a state revenue department of your choice or the state assigned to you by the instructor. Links to all state revenue departments can be...
-
An observer moving toward Earth with a speed of 0.85c notices that it takes 5.0 min for a person on Earth to fill her car with gas. Suppose, instead, that the observer had been moving away from Earth...
-
For the HiTech bond of Example 17.4, suppose that default is recognized only at maturity and that no restitution is made, that is, the default is ignored. What is the value of that bond? - Example...
-
1. If you were representing the Company in this case, what argument (facts and reasons) could you make that the confidentiality agreement had a legitimate business purpose and was applied...
-
according to the information that given below, what is the function that insert formulates (lookup-vlookup)to calculate employee superannuation 1 first_date fx 1/7/2020 H I J K L M N 0 P a 57...
-
A productivity index of 110% means that a company's labor costs would have been 10% higher if it had not made production improvements. Assume that Baldwin had a productivity index of 112% and that...
-
Explain how IR spectroscopy could be used to distinguish between thesecompounds: and b) and NH2 d d) and and e) ) and
-
Explain which functional group(s) is present in the compound that has this IRspectrum: 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (em)
-
If the public anticipates having to pay the future taxes implicit in any deficit, why will a lower tax not increase consumption spending (and aggregate demand)?
-
Six gallons of a salt solution consists of 35 % salt. It is the result of mixing a 40 % solution with a 30 % solution. How many gallons of each of the solutions was used? Let x = the number of...
-
When a car moves on a flat circular road at a constant speed of 15.0 m/s, the driver experiences a radial force of 120 N. What is the radial force on the driver if the speed of the car on this...
-
How much work would a child do while pulling a 12-kg wagon a distance of 4.3m with a 22N force?
-
Let S be the set that contains the smallest 100 positive integers; that is, S = {1, 2, 3, ... , 99, 100}. Prove that a list can be made of the 2100 subsets of S so that the empty set is the first...
-
a u au at x 0 0, u(x, 0) = 0, h=1/12 If then U,1 = O = O U,1 = U,1 = 1/2 (U+ 10 + U4-10) + 1/1 4 O Ui, 1 subjected to the conditions (x, 0) = 1 2 (U+1,1 + U-1,1)+ = 1/1 (14+10 + 24-10) i+ 2 at 1 U,1...
-
The project manager spends a great deal of her time communicating with project stakeholders. Which of the following represent an example of a stakeholder group for her project? a. Top management b....
-
Why are stocks usually more risky than bonds?
-
In the past, television commercials promoting toothpaste featured a well-known chef expressing satisfaction with the latest in toothpaste flavors, including citrus, herbal, mint, and cinnamon. U.S....
-
Evidence for the role of acetate in fatty-acid biosynthesis comes from isotope-labeling experiments. If acetate labeled with 13C in the methyl group (13CH3C02H) were incorporated into fatty acids at...
-
Does the reduction of acetoacetyl ACP in step 6 occur on the Re face or the Si face of themolecule? NADPH NADP+ SACP SACP H3C Acetoacetyl ACP B-Hydroxybutyryl ACP
-
Identify the two steps in glycolysis in which ATP is produced.
-
Determine the material inventory balance at the end of may? Received Issued Receiving Received Materials Report Number Received Quantity Unit Price Requisition Number Issued Quantity Issued Balance...
-
During October 2 0 2 3 , Fern Field Farms, Inc. received $ 1 0 , 0 0 0 from customers in exchange for fruit and vegetables. During the same month, the company paid $ 2 , 0 0 0 to employees, $ 5 0 0...
-
Given data below answer the question. Cash Accounts receivable $ 10,200 Cash dividends 15,200 Consulting revenue Office supplies 3,550 Rent expense $ 2,340 15,200 3,910 Office equipment 18,310 Land...

Study smarter with the SolutionInn App


