Explain which species is the stronger base: a) :CH or :CH NO b) CH-P-CH, or CH-N-CH CH3
Question:
Explain which species is the stronger base:
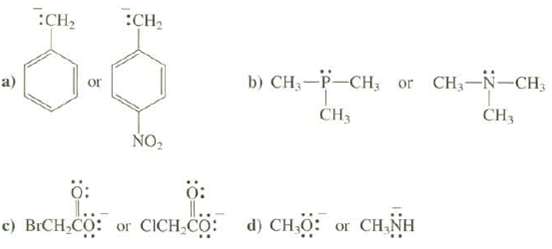
Transcribed Image Text:
a) :CH₂ or :CH₂ NO₂ b) CH₂-P-CH, or CH₂-N-CH₂ CH3 CH3 Ö: Ö: c) BICH,CO: or CICH,CO: d) CH₂O: or CH₂NH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a The anion on the left is a stronger base because the n...View the full answer

Answered By

Rajesh Das
A young enthusiastic researcher, blogger, and programmer currently pursuing an Integrated Master of Science in Applied Physics. I am working as a Physics Expert in Study.com (e-learning site). Also for the past 5 years, I have been providing personalized tutoring to students who are preparing for higher studies and competitive exams. I have a very good grasp on Quantum mechanics, Classical mechanics and Electronics. I started tutoring in 2016 and I have tutored students varying from class 9th to class 12th.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which species is a stronger acid? a) HS or HCI + b) PH or NH c) CHCH3 or HS
-
The compound shown is a somewhat stronger base than ammonia. Which nitrogen do you think is protonated when it is treated with an acid? Write a structural formula for the species that results. Refer...
-
Which species is more stable? a. b. c. d. e. f. CH10. CHOH,or or CH3CCHCH2CH or CH CCHCCH CH3CHCH2CCH3 or CH3CH CHCCH3 NH CHICHCHsor CHCNH CH CHCH3 or CHCNH2 CH2 CH C-CH or CH C-CH CH3 CH3 or
-
What are the costs of healthcare, where does the money come from, and where is it spent?
-
Rural Support Ltd has been successful in gaining a major contract to prepare and deliver meals to elderly and disabled people who reside in Windsor. These operations were previously managed by the...
-
In a pharmacological experiment, 35 lab rats were not given water for 11 hours and were then permitted access to water for 1 hour. The amounts of water consumed (mL/hour) are given in the following...
-
Describe the Waterfall Development Model, its characteristics, and shortcomings.
-
Bailey and Tepe, attorneys-at-law, provided legal representation to Goode Equipment Inc. in a product liability suit. Twenty partner hours and 65 associate hours were worked in defending the company....
-
Units Unit Cost Total Cost 1/1 Beginning Inventory 126 $7 $882 1/20 Purchase 504 $8 4,032 7/25 Purchase 252 $9 2,268 10/20 Purchase 378 $10 3,780 1,260 $10,962 A physical count of inventory on...
-
The Golden Goals, a professional soccer team, prepares financial statements on a monthly basis. The soccer season begins in May, but in April the team engaged in the following transactions: 1. Paid...
-
Explain which compound is the stronger acid: O a) CHCNH or CH3COH c) CHCHCHCH3 or e) CHNH or CH-NH b) CH3 SH or OH OH 6.6. & or d) CF3 CH3
-
Arrange these compounds in order of increasing acid strength: CHOH CN CN
-
In Problem find y in two ways: (A) Differentiate the given equation implicitly and then solve for y. (B) Solve the given equation for y and then differentiate directly. 4x 2 - e y = 10
-
You are given a sheet of fibrous connective tissue to investigate. The tissue fibers run in a single direction. When subjected to biaxial testing of forces f and f2, the tissue 1 undergoes a...
-
A beam is composed of two channels, C 811.5, as shown in the following figure. The beam is supported at the left end by a in- round eye bar. A % in pin is used at each eye. The beam is supported at C...
-
Determine the temperatures at which the values of Vrms, , and Vmp for CH4 (considered to be a gas of point-particles) become supersonic. (Supersonic velocities are larger than Mach-1 = 346.27 m/s in...
-
A balloon having a volume of 10,000 cu ft is filled with hot air at atmospheric pressure and a temperature of 280 F. If the temperature of the surround- ing air is 77 F and the weight of the balloon...
-
Communications satellites are often put into geosynchronous orbit (GEO). This is an orbit that keeps the satellite over a fixed point on the equator. The rocket launches the satellite into a...
-
Assume Stassen Company on January 1, 2017, decides to contract with another company to preassemble a large percentage of the components of its telescopes. The revised manufacturing cost structure...
-
A company pledges their receivables so they may Multiple Choice Charge a factoring fee. Increase sales. Recognize a sale. Collect a pledge fee. Borrow money. Failure by a promissory notes' maker to...
-
Prove each equation. (a) Equation 14a (b) Equation 14b 14a 14b tan(x + y) = tan(x - y)= = tan x + tan y 1 - tan x tan y tan x - tan y 1+tan x tan y
-
How would you synthesize the following compounds from Cyclohexanone? (a) 1-Methylcyclohexane (b) 2-Phenylcyclohexanone (c) cis-1, 2-Cyclohcxanediol (d) 1-Cyclohexylcyclohcxanol
-
One of the steps in the metabolism of fats is the reaction of an unsaturated acyl CoA with water to give a -hydroxy-acyl CoA. Propose a mechanism.
-
The amino acid methionine is biosynthesized by a multistep route that includes reaction of an imine of pyridoxal phosphate (PLP) to give an unsaturated imine, which then reacts with cysteine. What...
-
Powell Company began the Year 2 accounting period with $18,000 cash, $60,200 inventory, $49,400 common stock, and $28,800 retained earnings. During Year 2, Powell experienced the following events:...
-
Consider the following matrix A: 1 A = -22 -22 3 Use MATLAB to find the eigenvalues of matrix A. The command in MATLAB is [V,D]=eig(A). Where V is the matrix of eigenvalues and D is the matrix of...
-
a. What is the profitability index for the set of cash flows if the relevant discount rate is 11 percent? (Do not round intermediate calculations and round your answer to 3 decimal places, e.g.,...

Study smarter with the SolutionInn App


