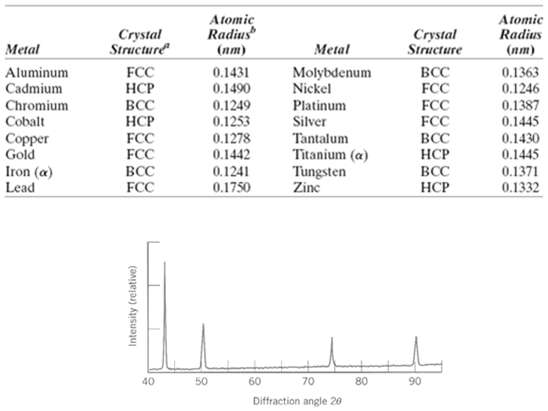
Figure shows the first four peaks of the x-ray diffraction pattern for copper, which has an FCC
Question:
Figure shows the first four peaks of the x-ray diffraction pattern for copper, which has an FCC crystal structure; monochromatic x-radiation having a wavelength of 0.1542 nm was used.
(a) Index (i.e., give h, k, and l indices) for each of these peaks.
(b) Determine the interplanar spacing for each of the peaks.
(c) For each peak, determine the atomic radius for Cu and compare these with the value presented in Table3.1.
Transcribed Image Text:
Atomic Radius Atomic Radius Crystal Structure Crystal Structure Metal Metal (пт) (пт) 0.1431 0.1490 BCC FCC Aluminum Cadmium FCC НСР Molybdenum Nickel 0.1363 0.1246 BCC НСР FCC FCC Platinum Silver Chromium Cobalt 0.1249 0.1253 0.1278 0.1442 FCC FCC 0.1387 0.1445 Tantalum Titanium (a) Tungsten Zine 0.1430 0.1445 0.1371 Copper Gold BCC НСР Iron (a) BCC 0.1241 BCC 0.1332 Lead FCC 0.1750 НСР 40 50 60 70 80 90 Diffraction angle 20 Intensity (relative)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a Since Cu has an FCC crystal structure only those peaks ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Figure 3.25 shows the first four peaks of the x-ray diffraction pattern for copper, which has an FCC crystal structure; monochromatic x-radiation having a wavelength of 0.1542 nm was used. (a) Index...
-
The metal iridium has an FCC crystal structure. If the angle of diffraction for the (220) set of planes occurs at 69.22 (first-order reflection) when monochromatic x radiation having a wavelength of...
-
The metal iridium has an FCC crystal structure. If the angle of diffraction for the (220) set of planes occurs at 69.22( (first-order reflection) when monochromatic x-radiation having a wavelength of...
-
Describe the impact of mobile computing upon your business and project its impact in the future. Do the same for social media. In both cases, when considering impacts, consider how each technology...
-
A small, dry paper clip can rest on the surface of still water. Why can't a heavier paper clip do the same without sinking?
-
What is at stake for the key parties? What are Donna's ethical obligations to them?
-
A double-pipe heat exchanger consists of two concentric tubes with one fluid flowing in the central tube and the other flowing in the annulus between the tubes. In a particular exchanger, cold...
-
Accounts Payable Confirmations. Partners Clark and Kent, both CPAs, are preparing their audit plan for the audit of accounts payable on Marlboro Corporations annual audit. Saturday afternoon they...
-
32) Refer to the drawing of an elevated centrifugal pump taking suction from the bottom of an open storage tank containing water at 66F (see figure below). Assume standard atmospheric pressure. The...
-
"Part 1: The Performance Lawn Equipment database contains data needed to develop a pro forma income statement. Dealers selling PLE products all receive 18% of sales revenue for their part of doing...
-
The diffraction peaks shown in figure are indexed according to the reflection rules for BCC (i.e., the sum h + k + l must be even). Cite the h, k, and l indices for the first four diffraction peaks...
-
Would you expect a material in which the atomic bonding is predominantly ionic in nature to be more or less likely to form a non-crystalline solid upon solidification than a covalent material? Why?
-
Find the orthogonal complement W ¥ of W and give a basis for W ¥ . : 3D 2t, t, z %3D 1 2
-
You have developed a chain of 10 cyber cafes near university campuses like Wits, UCT, UJ, Rhodes and Stellenbosch. Your business concept is to serve Java to the budding revolutionaries, the caviar...
-
Superior Sales Limited has share capital of $ 2 0 m as at 3 1 December 2 0 1 7 . Included in this share capital is $ 4 m preferred stocks. The company authorized share capital was 6 million shares...
-
1. Why and how do transaction costs in financial markets necessarily give rise to Financial Intermediaries? Explain. 2. What is the Lemons problem? Discuss 2 ways that the problem can be reduced.
-
The Birgini Company buys one unit of inventory for $ 7 7 in cash. This item is later sold for $ 1 0 9 on credit. What journal entry or entries are made at the time of sale if a periodic inventory...
-
A company has a $36 million portfolio with a beta of 1.2. The futures price for a contract on an index is 900. Futures contracts on $250 times the index can be traded. What trade is necessary to...
-
Consider a. Evaluate \(\dot{x}_{s s}\) by using the final-value theorem. b. Confirm the result of (a) by evaluating \(\lim _{t ightarrow \infty} \dot{x}(t)\). 0.2s+0.3 X(s) = s(3s +2.5s+0.5)
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
A jewelry designer plans to incorporate a component made of gold in the shape of a frustum of a cone of height 1 cm and fixed lower radius r (Figure 43). The upper radius x can take on any value...
-
Following is tabulated data that were gathered from a series of Charpy impact tests on a ductile cast iron. (a) Plot the data as impact energy versus temperature. (b) Determine a ductile-to-brittle...
-
Following is tabulated data that were gathered from a series of Charpy impact tests on a tempered 4140 steel alloy. (a) Plot the data as impact energy versus temperature. (b) Determine a...
-
A fatigue test was conducted in which the mean stress was 50 MPa (7250 psi) and the stress amplitude was 225 MPa (32,625 psi). (a) Compute the maximum and minimum stress levels. (b) Compute the...
-
Suppose you invest 52%, 28%, and 20% of your wealth into a stock, the market, and a risk-free asset, respectively. The beta of the stock is 1.1. What is the beta of the portfolio?
-
An investment of $21745, earning compound interest, grows by $2278 in one year. At this rate of growth, how long will it take for the original investment to double?
-
On September 12, 3,400 shares of Denver Company's common stock are acquired at a price of $58 per share plus a $170 brokerage commission. On October 15, an $1.20-per-share dividend was received on...

Study smarter with the SolutionInn App


