Find the steady-state response vo(t) for the network infigure. 12 cos t V 1 F 1F (1)a
Question:
Find the steady-state response vo(t) for the network infigure.
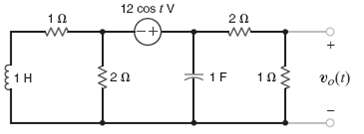
Transcribed Image Text:
12 cos t V 1 F 1F (1)°a 2. w-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
jta ord IN cas ut phases 12 0 3202 Z 21 4 4 7 2 12 3j1 m j 12 10 ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Electrical Engineering questions
-
Find the steady state response Vo (t) for the network in fig.
-
Find the steady state response Vo (t) for the network in fig. Discuss.
-
Find the steady state response Io (t) for the network shown the fig.
-
Reference groups influence us in three ways. These influences include informational, utilitarian, and dimensions. a. knowledge O b. O c. reputational O d. descriptive value-expressive
-
If a mass m is attached to a given spring, its period of oscillation is T. If two such springs are connected end to end and the same mass m is attached, (a) Is the resulting period of oscillation...
-
KNB sold real property to Firm P for $15,000 cash and Firm Ps assumption of the $85,000 mortgage on the property. a. What is KNBs amount realized on sale? b. Compute KNBs after-tax cash flow from the...
-
What is meant by the term fact pleading?
-
On January 1, 20X1, Wade Crimbring, Inc., a dealer in used manufacturing equipment, sold a CNC milling machine to Fletcher Bros., a new business that plans to fabricate utility trailers. To conserve...
-
How do leaders respond to ethical failures, breaches of trust, and reputational crises, demonstrating humility, accountability, and a commitment to organizational learning and continuous improvement,...
-
The design of a pump and pipe system has been completed, except for the valves. The system is used to transport water at 120F through 2 nom sch 40 commercial steel pipe at a required flow rate of 85...
-
Find the steady-state response vo(t) for the circuit shown infigure. 10 cos t A ww 10 10 vo(1) 1H 2.
-
Determine the steady-state response vo(t) for the network infigure. (1)'a 4 cos 21 V vo(t) 2v1(t) -/-
-
List some changes introduced by IoT.
-
The analysis performed by Sardar on the Purity Fund can be best described as being based on: A. a holdings-based approach. B. manager self-identification. C. a returns-based style analysis. Jack...
-
The arrival cost for purchasing the 90,000 shares of BYYP is: A. 164.4 bp. B. 227.2 bp. C. 355.0 bp. Robert Harding is a portfolio manager at ValleyRise, a hedge fund based in the United States....
-
Based upon Deweys chosen investment process for the management of the Purity Fund, rebalancing of the fund will most likely occur: A. at regular intervals. B. in response to changes in...
-
As it relates to the trade policy document, ValleyRise should implement Yellows recommendation related to: A. the list of eligible brokers. B. a policy for the treatment of trade errors. C. a policy...
-
Given her investment goals and market view, and assuming all options are based on SEK/CHF, the best strategy for Bjrk to manage the funds CHF exposure would be to buy an: A. ATM call option. B. ITM...
-
What is malware, and how does a computer virus work?
-
3.16. For a system with non-identical service rates (see Sect. 3.5) and a limit of N jobs in the system (Eq. 3.13), obtain an expression for the mean service time per job, E[Ts], as a function of the...
-
Why are nonmetals important even though they account for only a very small fraction of the elements in the periodic table?
-
An ideal gas having a constant specific heat undergoes a reversible polytropic expansion with exponent, n = 1.4. If the gas is carbon dioxide will the heat transfer for this process be positive,...
-
A cylinder/piston contains 1 kg methane gas at 100 kPa, 20C. The gas is compressed reversibly to a pressure of 800 kPa. Calculate the work required if the process is a. Adiabatic b. Isothermal c....
-
Helium in a piston/cylinder at 20C, 100 kPa is brought to 400 K in a reversible polytropic process with exponent n = 1.25. You may assume helium is an ideal gas with constant specific heat. Find the...
-
Suresh Company reports the following segment (department) income results for the year. Sales: Expenses Avoidable Unavoidable Total expenses Income (loss) Department M $ 66,000 Department N $ 38,000...
-
Question : List the competitive advantages of Nordstrom: the things that make it different from competitors in positive ways. Nordstrom: A Shining Example of Service Excellence 'Use your best...
-
EZ Tees Corporation produces T-shirts. The company uses a standard costing system and has set the following standards for direct materials and direct labor (for one shirt): Fabric (1.5 yds. @ $2.80)...

Study smarter with the SolutionInn App


