Find vo(t), for t > 0, in the network infigure. 2H 60 80 ww F 4H {8H
Question:
Find vo(t), for t > 0, in the network infigure.
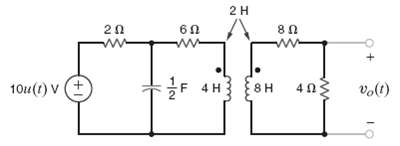
Transcribed Image Text:
2H 60 80 ww F 4H {8H 10u(1) V 8H 403 (1)°a (+1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
t0 No voltage applied All Initial conditions ane gero 20 Vocs 10 v 21s ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Electrical Engineering questions
-
Find vo(t), for t > 0, in the network infigure ww 2 kN w- 2 kn C 4 kn 3 kn 8 kn 2 kn 100 F 24 V 34 kn v.(t) 4 kn +) 12 V
-
Find V0(t) for t > 0 in the network in Fig. P6.32 using the step-by-step technique. 200 F 12V /+ t= 0
-
Find VC(t) for t > 0 in the network in Fig. 6.25 using the step-by-step method. Volt) 100 100 F - 12V
-
Process compliance can be affected by: Multiple select question. organizational structure. maverick purchasing. organizational culture. information systems
-
An observant fan at a baseball game notices that the radio commentators have lowered a microphone from their booth to just a few inches above the ground, as shown in FIGURE 13-36. The microphone is...
-
Discuss why and how a researcher might use secondary authorities in performing tax research.
-
What must the respondent do to oppose a motion?
-
The following table shows some simple student data as of the date 06/20/2015: The following transactions occur on 06/21/2015: Student 004 changes major from Math to Business. Student 005 is deleted...
-
What can companies do to make sure their employees are aware of the consequences of financial securities fraud? Provide several examples.
-
Find the simple interest for each of the following. Round to the nearest cent. 1. $27,850 at 7.75% for 100 days 2. $7800 at 9.25% for 4 months 3. A loan of $5800 at 9% made on May 22 and due July 5...
-
Find vo(t), for t > 0, in the network infigure. ww ww 4u(1) v(+ vo(t) 2H 2 H
-
Find vo(t), for t > 0, in the network infigure. 1:2 ww 4 0 1 F 12u(1) V vo(t) + Ideal -/4
-
The accompanying scatter diagram shows the numbers of hours several students studied and their midterm scores. (a) Which is the dependent variable and which the independent variable? (b) Is there a...
-
Adidas provoked outrage when the company posted a photo of a new design, the JS Roundhouse Mids, on its Facebook page. The shoes come with bright orange shackles and the tagline Got a sneaker game so...
-
Develop the appropriate primary research question to be associated with this design. Develop a hypothetical research scenario that would necessitate the use of a Longitudinal Design. The research...
-
A system at constant pressure consists of \(10 \mathrm{~kg}\) of air at a temperature of \(1000 \mathrm{~K}\). This is connected to a large reservoir which is maintained at a temperature of \(300...
-
In the context of the model introduced in Kyle [1147] and discussed in Sect. 10.2, let the couple \((X, P)\) represent the linear equilibrium, as stated in Proposition 10.3. Prove the following...
-
Reconsider Problem 11. Plot a graph that shows the planning horizon as the independent variable and the required deposit as the dependent variable. Plot planning horizons from 1 to 250 with a step...
-
Why DOE is necessary for chemical process design?
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Tool Manufacturing has an expected EBIT of $57,000 in perpetuity and a tax rate of 21 percent. The firm has $134,000 in outstanding debt at an interest rate of 5.35 percent and its unlevered cost of...
-
Determine the missing property among P, T, s, x for the following states: a. Ammonia 25oC, v = 0.10 m3/kg b. Ammonia 1000 kPa, s = 5.2 kJ/kg K c. R-134a 5oC, s = 1.7 kJ/kg K d. R-134a 50oC, s = 1.9...
-
Consider a Carnot-cycle heat engine with water as the working fluid. The heat transfer to the water occurs at 300C, during which process the water changes from saturated liquid to saturated vapor....
-
In a Carnot engine with ammonia as the working fluid, the high temperature is 60C and as QH is received, the ammonia changes from saturated liquid to saturated vapor. The ammonia pressure at the low...
-
Please answer all parts of the following question. Given the following information: Total Product (TP) Price Fixed Cost (TFC) Variable Cost (TVC) 0 $1000 $600 $ 0 1 1000 600 1200 2 1000 600 2000 3...
-
Daniel made the following transactions during the first week of January. 1 January Buys stock for 350 on credit 3 January Makes sales of 740 on credit 4 January Cash paid to a supplier of 120, for...
-
Imagine you are an analyst working at a financial consulting firm. Your organization has been hired by UBS to provide advice following a major legal and ethical crisis. You have been asked to draft...

Study smarter with the SolutionInn App


