Forty-five kilograms of a solution containing 30 wt% ethylene glycol in water is to be extracted with
Question:
Forty-five kilograms of a solution containing 30 wt% ethylene glycol in water is to be extracted with furfural. Using Figures 4.14a and 4.14c, calculate:(a) The minimum quantity of solvent.(b) The maximum quantity of solvent.(c) The weights of solvent-free extract and raffinate for 45 kg solvent, and the percent glycol extracted.(d) The maximum possible purity of glycol in the finished extract and the maximum purity of water in the raffinate for one equilibriumstage.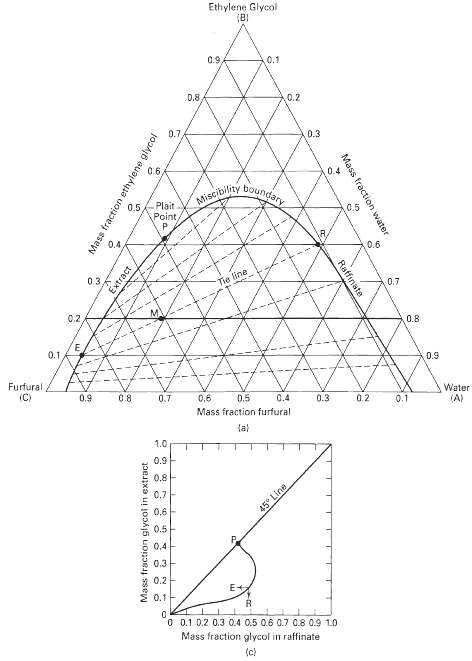
Transcribed Image Text:
Ethylene Glycol (B) 0.9, 0.1 0.8, 0.2 0.7 0.3 0.4 0.6 Miscibity boundary Plait 0.5 0.5 Point, 0.4 0.6 0.3 0.7 Tie line 0.2 0.8 0.1. 0.9 Furfural (C) Water (A) 0.2 0.9 0.8 0.7 0,6 0.5 0.4 0.3 0.1 Mass fraction furfural la) 1.0 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 O 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1.0 Mass fraction glycol in raffinate (c) Mass fraction ethylene glycol Mass fraction water Raffinate 45° Line Mass fraction glycol in extract
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
For a single stage all mixtures of feed F and solvent S lie on a straight line between these two points as shown in the following ternary diagram of F...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A quantity of 25.0mL of a solution containing both Fe2+ and Fe3+ ions is titrated with 23.0mL of 0.0200 M KMnO4 (in dilute sulfuric acid). As a result, all of the Fe2+ ions are oxidized to Fe3+ ions....
-
Using activities, calculate the pH of a solution containing 0.010M NaOH plus 0.0120 M LiNO3. What would be the pH if you neglected activities?
-
Water is to be boiled at sea level in a 30-cm-diameter stainless steel pan placed on top of a 3kW electric burner. If 60 percent of the heat generated by the burner is transferred to the water during...
-
What does the following code fragment print? int[] a = { 1, 2, 3 }; int[] b = { 1, 2, 3 }; System.out.println(a == b);
-
Canam Group Inc.'s receivables turnover was 3.7 times in 2010 and 6.3 times in 2009. Has Canam's receivables management improved or worsened?
-
On December 12, 2018, Pace Electronics received $24,000 from a customer toward a cash sale of $240,000 of diodes to be completed on January 16, 2019. What journal entries should Pace record on...
-
The triangular plate ABC is deformed into the shape shown by the dashed lines. If at A, AB = 0.0075, AC = 0.01 and xy = 0.005 rad, determine the average normal strain along edge BC. C y 300 mm...
-
Now assume that a court finds that the new rule is merely interpretive. What effect would this determination have on whether the SEC had to follow the APAs rulemaking procedures? Assume that the...
-
Two resistors are connected in parallel with an 99 volt battery. The resistors have values of R 1 = 7 ohms and R 2 = 55 ohms. What is the current (in amps) in this circuit? Round your answer to two...
-
as || , you will design a simple salad bar interface. The program will display a main menu to the user to begin. User will choose from given options to order types of salad, calculate and display...
-
A feed of 13,500 kgh consists of 8 wt% acetic acid (B) in water (A). The removal of the acetic acid is to be accomplished by liquid-liquid extraction at 25oC. The raffinate is to contain only 1 wt%...
-
Prove that, in a triangular diagram, where each vertex represents a pure component, the composition of the system at any point inside the triangle is proportional to the length of the respective...
-
Manu Narayan owns several first-class hotels worldwide, including properties in Manhattan, Mumbai, and even some in suburbia. He wants to make sure that the humancomputer interface is appropriate to...
-
Carol purchased a holiday flat in December 2007 for 100,000. She spent 5,000 on installing central heating in February 2008 and a further 750 on repainting the interior of the flat in March 2008. The...
-
In January 2018 Karl sells a one -quarter interest in a chattel for 2,500. On the date of this sale, the remaining three -quarters interest is valued at 8 ,500. The chattel had cost Karl 3,850 in...
-
Rosemary's capital gains and losses in recent years (and the annual exemption for each year) have been as follows: There were no unrelieved losses to bring forward from 2013 -14 or earlier. Compute...
-
Which of the following are chargeable assets for CGT purposes? (a) shares in a limited company (b) gilt-edged securities (c) an antique table worth 20,000 (d) an antique chair worth 5,000 (e) a...
-
Irma is self-employed. Her trading income for 2017 -18 is 64,880 and during the year she makes net contributions to a registered pension scheme of 6,000. Her only other income in 2017-18 consists of...
-
Wilma purchased a used minivan, including a three-month warranty on parts and labour, from Xenon Used Cars Inc. for $10 000. She paid $1500 down and signed a promissory note for $8500, payable in...
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
Is talent contextual?
-
Separation of air into N 2 and O 2 is widely practiced. Cryogenic distillation is most economical for processing 100 to 5,000 tons of air per day, while pressure-swing adsorption is favorable for 20...
-
Beginning with the Ruth equation (14-24), obtain general expressions for time-dependent permeate volume, V{t}, and timedependent flux, J{t}, in terms of operating parameters and characteristics of...
-
What is varied to achieve optimal control?
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
Marcia Stanton is the new manager of the materials storeroom for Taylor Manufacturing. Marcia has been asked to estimate future monthly purchase costs for part #696, used in two of Taylor's products....
-
Miller Company's contribution format income statement for the most recent month is shown below: Sales (37,000 units) Variable expenses Contribution margin Fixed expenses Net operating income...

Study smarter with the SolutionInn App


