Galactose, a constituent of the disaccharide lactose found in dairy products, is metabolized by a pathway that
Question:
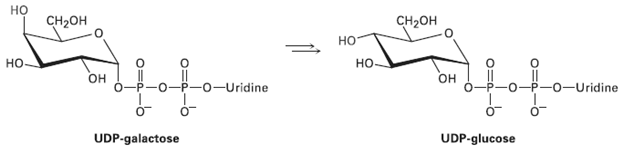
Galactose, a constituent of the disaccharide lactose found in dairy products, is metabolized by a pathway that includes the isomerization of UDP-galactose to UDP-glucose, where UDP = uridylyl diphosphate. The enzyme responsible for the transformation uses NAD+ as cofactor. Propose a mechanism.
Transcribed Image Text:
но CH-он CH2он но но но -Uridine он -0-Uridine н UDP-galactose UDP-glucose
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
Base NAD H HO CHOH OH UDPGalactose UDP H NADH HO CHOH OH UDP HO ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The disaccharide lactose can be decomposed into its constituent sugars galactose and glucose. This decomposition can be accomplished through acid-based hydrolysis, or by the enzyme lactase. Lactose...
-
Propose a mechanism for methylmalonyl-CoA mutase, the enzyme that converts methylmalonyl-CoA into succinyl-CoA.
-
Glucose is the main fuel metabolized in animal cells: C6H12O6 + 6O2 ( 6CO2 + 6H2O H = 2,799 kJ How much energy is given off when 100.0 g of C6H12O6 react?
-
A random walk in an undirected connected graph moves from a vertex to one of its neighbors, where each possibility has equal probability of being chosen. (This process is the random surfer analog for...
-
Name five potentially valuable indicators of a firm's social responsibility and describe how company performance in each could be measured.
-
a) If total spending increases, or decreases, does this create a tradeoff between un-employment and inflation? b) What could cause a simultaneous decrease in unemployment and inflation? What could...
-
What accounting control principles are applicable to the recording of cash receipts and sales adjustments transactions?
-
For its top managers, Worldwide Travel formats its income statement as follows: Worldwide's relevant range is between sales of $245,000 and $364,000. Requirements 1. Calculate the contribution margin...
-
1) The December 31, 2024, balance sheet of Garcia Company included the following: Common stock, 20 million shares outstanding at $1 par Paid-in capital-excess of par Retained earnings $20,000,000...
-
The income statements for Paste Company and its subsidiaries, Waste Company, and Baste Company were prepared for the year ended December 31, Year 6, and are shown below: Additional Information ¢...
-
Identify the reagents a?f in the following scheme: .Br CH-
-
Propose a structure consistent with the following spectral data for a compound C8H18O2: IR: 3350 cm1. 1H NMR: 1.24 (12 H, singlet); 1.56 (4 H, singlet); 1.95 (2 H, singlet)
-
The graph is a dotplot of the number of pairs of shoes owned by men and women who took a survey on StatCrunch. a. Shape: What is the shape of each dotplot? b. Center: Is it the males or the females...
-
. Discuss how is the top-down approach to information security superior to the bottom-up approach. Which approach was used in your organization? 2. Can recent college graduates expect to be project...
-
A baseball bat has 120 J of kinetic energy as it hits a baseball (m = 0.145 kg). (a) If the baseball bat transfers 70 J of its kinetic energy to the baseball, what is the efficiency of the energy...
-
If f(x) = (x + 1)(2-3x), then f(1) =
-
Identify a recent interpersonal, group, or organizational conflict that you were involved in and was later resolved. Select five of the topics listed below and discuss how they apply to your chosen...
-
In the context of cellular respiration, what molecular mechanisms regulate the synthesis and utilization of ATP, the primary energy currency of cells, and how do respiratory substrates like glucose...
-
When a contracting hot ball of gas spins into a disk shape, it cools faster due to (a) increased radiation transfer. (b) increased surface area. (c) decreased insulation. (d) increased convection...
-
Assume that a trial balance is prepared with an account balance of $21,360 listed as $21,630 and an account balance of $1,500 listed as $15,000. Identify the transposition and the slide.
-
Surface tension, viscosity, and vapor pressure are all related to intermolecular forces. Why do surface tension and viscosity decrease with temperature, whereas vapor pressure increases with...
-
1-Bromo [2.2.1] bicycloheptane is unreactive toward both SN2 and SN1 reactions. Open the computer molecular model at the book's website titled "1-Bromo [2.2.1] bicycloheptane" and examine the...
-
Open the computer molecular model titled "1-Bromo [2.2.1] bicycloheptane LUMO" at the book's website for the lowest unoccupied molecular orbital (LUMO) of this compound. Where is the lobe of the LUMO...
-
In the previous problem and the associated molecular model at the book's website, you considered the role of HOMOs and LUMOs in an SN2 reaction. (a) What is the LUMO in an SN1 reaction and in what...
-
I borrowed $21,500 for 6 years at 8.48%. Assuming that I make all of my monthly payments as scheduled, how much will I owe at the end of 3 years?
-
Comparing Net Present Value with Internal Rate of Return 1. Starting from the Excel spreadsheet and using the =IRR(values,[guess]) Excel function, calculate the Internal Rate of Return (IRR) for...
-
Hedging EUR currency risk from the US perspective Terabit Inc is an American exporting company of electronic components to countries in the Euro area. The company mainly sells its products in Euro,...

Study smarter with the SolutionInn App


