Ultraviolet spectroscopy of often used to monitor the amount of a protein in a sample, the amount
Question:
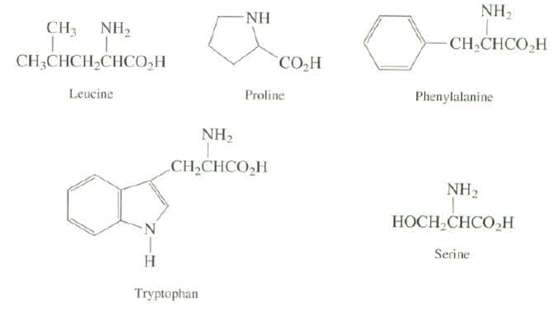
Ultraviolet spectroscopy of often used to monitor the amount of a protein in a sample, the amount of protein is correlated with the absorbance at 280nm. Explain which of the following amino acids you expect to have an absorption at 280nm, (the carbonyl groups of the peptide [amide] bond of the protein do not absorb at thiswavelength).
Transcribed Image Text:
NH, NH NH, CH3 CH CHCO,H CH,CHCH,CHCO,H CO,H Leucine Proline Phenylalanine NH2 CH,CHCO,H NH, НОСН-СHCO,H Serine H. Tryptophan
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (8 reviews)
To have an absorption at 280 nm the amino acid m...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain how FRET could be used to monitor the association of G"' and adenylyl cyclase following activation of the epinephrine receptor.
-
Which of the following amino acids has a nonpolar side chain? NH2 CHSCH2CH2CH COOH NH2 HSCH CH COOH
-
Which of the following amino acids are likely to be linked to only one species of tRNA? What are their anticodons? (a) Phe (b) Leu (c) His
-
Let y be a variable that takes 0 or 1 value. Write an inequality to describe if y = 0, then x can take value from 0 to 5 and if y = 1, then x can take value 0. We are given 3 options. We are allowed...
-
Sales have decreased for two consecutive quarters at your business. You have been instructed to inform your sales team that their hours, and base pay, will be reduced by 20 percent. While you may...
-
What are some of the actions senior leaders can take to show they are committed to long term company success?
-
For each of the following, calculate the standard error of the difference \(\left(s \mathrm{X}^{-} 1-\mathrm{X}^{-} ight.\) 2 ) and the \(t\)-test for independent means. a. N = 6, N = 6, X 1 18.50, $...
-
The financial statements of P&G are presented in Appendix B. The companys complete annual report, including the notes to the financial statements, is available online. Instructions Refer to P&Gs...
-
Create a Assignment: Implementing Lean Operations Assume that you are a manager at a small coffee shop or a McDonald's fast-food restaurant. Your task is to implement lean operations to improve...
-
Determine the principal stress at point A on the cross section of the hanger at section a-a and b-b. Specify the orientation of the state of stress and indicate on the element at the point. Draw...
-
The 1H-NMR spectrum of compound H is as follows, the peaks at highest m/z in the mass spectrum of H appear at 122 and 124 with intensities in a ration of about 1 to 1. Show the structure ofH. 2 H ...
-
Explain why the ultraviolet spectrum of one of these dienes has its maximum absorption at a longer wavelength than that of the other.
-
In one group, find a local business, such as a copy shop, that charges for printing, faxing, copying, and scanning documents. In the other group, determine the price of a mid-range...
-
Write a Java function that efficiently calculates the edit distance between two strings.
-
Make a Contact Us page as given in the following link : (HTML Doc) https://www.tune.com/contact-us/ You can skip the design part. However, the layout and content should be same. Do the Header and...
-
Write a program that takes a string representing a mathematical expression and returns the result of evaluating the expression. Support basic arithmetic operations and parentheses.
-
Image transcription text . What is your perception of Whole Foods Market? . From a consumer standpoint, do you see the appeal of the 365 by Whole Food Market concept? . . While the 365 concept was...
-
calculate the following for all 3 years. 1-Net operating working Capital 2-Working Capital 3-Net Working Capital 6703 8 9 Consolidated Scale Cash & equivalents Accounts & notes receivable 14...
-
A small manufacturer has won a major contract with the U.S. Army to develop a new generation of satellite phone for battlefield applications. Because of the significant technological challenges...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
In 2008, the financial markets and banking system of the United States nearly collapsed completely as a result of the issuance and securitization of subprime mortgages. When the housing market began...
-
What is the conjugate acid of each of the following bases? (a) HSO4- (b) H2O (c) CH3NH2 (d) -NH2 (e) CH3CH2 (f) CH3CO2-
-
Designate the Lewis acid and Lewis base in each of the following reactions: (a) (b) (c) CI CH,CH2 CIAIC CH OHBFCH3 CH3 CHs CH
-
Rewrite each of the following reactions using curved arrows and show all nonbonding electron pairs: (a) (b) (c) CH NH2 HC
-
Ned needs to produce more of his two products in order to increase his net income and has found a way to increase the limited resource, machine hours. He has two products, hammers and screwdrivers.....
-
What effect does an antagonist drug have over the receptors ?
-
Read this article on the differences and Comparisons between UNIX and LINUX http://www.diffen.com/difference/Linux_vs_Unix, then do some research and post your thoughts on each of the systems. Based...

Study smarter with the SolutionInn App


