Give IUPAC names for the followingcompounds: (a) Br (b) (c) CH CH3CH2CHCH2CH2CH3 CHH-CH2Co CHCH2C
Question:
Give IUPAC names for the followingcompounds:
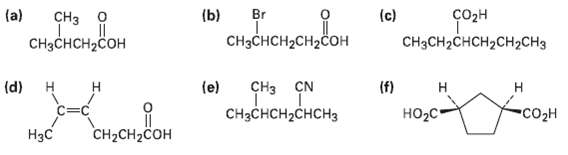
Transcribed Image Text:
(a) Br (b) еке (c) соон CHз о CH3CH2CHCH2CH2CH3 CHзснсH-CH2Coн CHзснCH2Cон (f) НO2с- (d) н CHз CN (e) н н Н CHзCнCH2CHсHз "Co2н CH2CH2COH Нзс
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Carboxylic acids are named by replacing e of the corresponding alkane with oic acid The carboxylic acid carbon is C1 WhenCOH is a substituent of a ring the suffix carboxylic acid is used the carboxyl ...View the full answer

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give IUPAC names for the following compounds. (a) (b) (c) CH3 CH CH CH CH, CH, CH CH
-
A proposed project has fixed costs of $84,000 per year. The operating cash flow at 7,500 units is $93,000. Ignoring the effect of taxes, what is the degree of operating leverage? If units sold rise...
-
Is New Belgium Brewery centralized or decentralized? How does this make them more effective?
-
The accompanying chart (Figure P8.6) shows the expected monthly profit or loss of Cypress Manufacturing Company within the range of its monthly practical operating capacity. Using the information...
-
Find the power spectral density of a doubly stochastic Poisson impulse process having a rate process \(\Lambda(t)\) described by \[ \Lambda(t)=\lambda_{0}[1+\cos (2 \pi \bar{v} t+\Phi)] \] where...
-
An aqueous solution containing 85.0 wt% H2SO4 at 60F (specific gravity = 1.78) is diluted with pure liquid water at the same temperature. The feed solution volume is 350 mL. The mixing may be...
-
Assume that an old-school horror movie spider is shaded like a sphere that is 30 feet in diameter. The spider needs 26 ml of oxygen per second per cubic centimeter. It can absorb 26.4 ml of oxygen...
-
Record all transactions in appropriate T-accounts (costs by function, LIFO perpetual), close all accounts, and calculate the ending balances. Opening balances: Material A: 100 units x 2,5 / unit =...
-
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs. CH- NABHA -2 Sorbitol Glucose
-
Draw structures corresponding to the following IUPAC names: (a) 2, 3-Dimethylhexanoic acid (b) 4-Methylpentanoic acid (c) Trans-1, 2-Cyclobutanedicarboxylic acid (d) o-Hydroxybenzoic acid (e) (9Z,...
-
What is the voltage of a concentration cell of Cl - ions where the concentrations are 1.045 and 0.085 M ? What is the spontaneous reaction?
-
What are the key elements to consider when designing a new product or service? How do you prioritize features in the design process for a product or service? Can you explain the importance of...
-
Durkheim noted that psychological explanations are insufficient when analyzing social behavior. In your own words, tell what this statement means?
-
Briefly list some points to note that can help when planning a wine list: 1) The list should have a logic that is easily understood by the client 2) Wines should be selected that are consistent in...
-
8. Specifics of real estate investment - Tax liability and leveraging Making Real Estate Investments Real estate has been a lucrative Investment for many years. Real estate provides greater...
-
What factors would you consider when deciding between in-person, online, or blended learning for a technical skills training program?
-
Consider the model proposed by Kyle [1148] and described in Sect. 10.1. By making use of the multivariate normality assumption on the random variables \(\left(\tilde{d}, \tilde{y}_{1}, \ldots,...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
How much heat (in kJ) is evolved in converting 1.00 mol of steam at 145 C to ice at -50 C? The heat capacity of steam is 2.01 J/g C, and that of ice is 2.09 J/g C.
-
Complete the following equations: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) Phenol + NaH (k) Product of (j) + CH3OSO2OCH3 (l) Product of (j) + CH3I (m) Product of (j) + C6H5CH2Cl PhenolBr2 C, CS2...
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
A synthesis of the b-receptor blocker called toliprolol begins with a reaction between 3-methylphenol and epichlorohydrin. The synthesis is outlined below. Give the structures of the intermediates...
-
The government standard on radiation from electrical devices is 10mW/cm2 (maximum). Assume a laptop computer is found to radiate more than this standard. The electric field inside is measured to be 5...
-
A 2 2 . 8 - m deep pool is filled with oil of density 8 9 5 kg / m ^ 3 . What is the gauge pressure at the bottom of the pool? Part B: What is the force from the okl that exerts on a 0 . 3 5 m x 0 ....
-
Clark was driving his four-wheeler 15 meters per second on a dirt road. It had just rained and there were massive mud puddles on the road. He hits on and it slows him to a speed of 3.7 meters per...

Study smarter with the SolutionInn App


