Give IUPAC names for the followingcompounds: H CHCH2CCH H CH CHH2CH2CH (b) (e) CHH2H2C CH
Question:
Give IUPAC names for the followingcompounds:
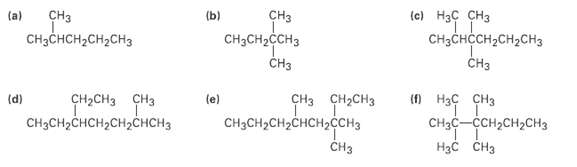
Transcribed Image Text:
сHз CHзCH2CCHз сHз CHз CHзснсH2CH2CHз (b) (e) Нзс сНз CHаснссH2сH2Cнз CHз (a) сH-сHз сHз CH3CH2CHCH2CH,CHCH3 (e) CHз снасHз CHзCH2CH2CHCH2сCHз CHз о Нас снз CHзс—ссн-сH>сH3 Нас снз (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
CH3 CH3CHCHCHCH3 2Methylpentane d CHCH3 CH3 CH3CHCHC...View the full answer

Answered By

Evans Cherono
I am an Information Technology Graduate and willing to work on any computer science or IT work to ensure I do my best all the time.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give IUPAC names for the following compounds. (a) (b) (c) CH3 CH CH CH CH, CH, CH CH
-
Add a toString() method to Sketch that returns a string representation of the unit vector corresponding to the sketch.
-
Suppose Shaan invested just $10,000 of his own money and had a $90,000 mortgage with an interest rate of 8.5 percent. After three years he sold the property for $120,000. a. What is his gross profit?...
-
Refer to the wage data given in Table 9-2 (found on the textbook's Web site). Regression (9.30) gives the results of the regression of the log of wage on the log of education. a. Based on the data of...
-
Listed below are a number of errors or irregularities pertaining to application controls in an EDP system: 1. An incorrect file is updated. 2. Output is distributed to unauthorized users. 3. Input...
-
Kathy Wintz formed a lawn service business as a summer job. To start the business on May 1, she deposited $1,000 in a new bank account in the name of the business. The $1,000 consisted of a $600 loan...
-
Wildhorse Corporation sells three different models of a mosquito "zapper. Model A12 sells for $65 and has unit variable costs of $46.00. Model B22 sells for $130 and has unit variable costs of...
-
Expand the following function in a Fourier series then find f (0), 6) f (z) =| x |, -5 < = < 0, >| | I am in a short exam, I want the solution as soon as possible
-
For each of the following compounds, draw an isomer that has the same functional groups. Each intersection of lines represents a carbon atom with the appropriate number of hydrogensattached. ( CH (a)...
-
Name the five isomers of C6H14. Discuss.
-
The annual demand for an item is 2400 units. The inventory holding cost is $ 6.00 per unit per year. The demand is continuous and constant, that is, 200 units/month. The item is purchased in two...
-
1) Consider an automated teller machine (ATM) in which users provide a personal identification number (PIN). Discuss what confidentiality, integrity, availability, authenticity, and non-repudiation...
-
Explain how Amazon might conduct a financial cost-benefit analysis of two day delivery. How would this financial cost-benefit analysis differ from an economic cost-benefit analysis? What costs and/or...
-
7. During the next 12 months, Crown, Inc. plans to pay dividends of $1.20 per share. Currently, its stock price is $30 per share. Growth is expected at 9 percent. Flotation costs are $2. Compute the...
-
Discuss the economic role of financial institutions and markets.
-
Find the PV of this 3-year annuity $100 ordinary annuity when the quoted interest rate is 4% compounded monthly
-
Natsam Corporation has \($250\) million of excess cash. The firm has no debt and 350 million shares outstanding, with a current market price of \($20\) per share. Natsams board has decided to pay out...
-
Use the T account for Cash below to record the portion of each of the following transactions, if any that affect cash. How do these transactions affect the companys liquidity? Jan. 2 Provided...
-
A 54 cm 3 ice cube at -25.0 C is added to a thermally insulated container with 400.0 mL H 2 O(l) at 32.0 C. What will be the final temperature in the container and what state(s) of matter will be...
-
What are the masses and structures of the ions produced in the following cleavage pathways? (a) A-cleavage of 2-methyl-3-hexanone (two pathways) (b) Dehydration of cyclopentanol (c) McLafferty...
-
Predict the masses and relative intensities of the peaks in the molecular ion region for the following compound. Br
-
Ethyl bromide and methoxybenzene (shown below) have the same nominal molecular weights, displaying a significant peak at m/z 108. Regarding their molecular ions, what other features would allow the...
-
What challenges arise when the HRM is uninformed concerning their legal rights and responsibilities? What is the potential impact on the organization? What should employees' expectations be...
-
Elaborate on "The Value of Learning of Applied Research Methods in Sports Management or Kinesiology" as another section of your Applied Research Methods in Sports Management. source:...
-
1. A entered into a contract with B, who is a minor. Can A later asks for the invalidation of the contract by reason of the minority of B? 10 points 2. A, the seller, and B, the buyer, agreed for the...

Study smarter with the SolutionInn App


