Give IUPAC names for the following substances (red = O, blue =N): (a) (b)
Question:
Give IUPAC names for the following substances (red = O, blue =N):
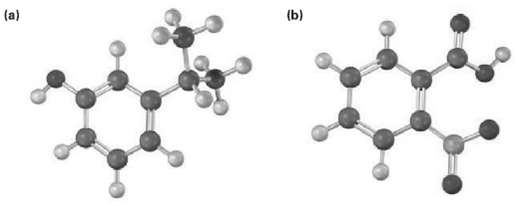
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a CH3 CHCH3 ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC names for the following alkenes: (a) (b) (c) (d) (e) (f) OH CI
-
Give IUPAC names for the following compounds. (a) (b) (c) (d) (e) (f) Ph CH3C C CH CH H3C CH3 - (CH3)3C C-CH (CH3)CH2CH3 CH CHC CC-OH CH,CH CH C-C CH
-
Give IUPAC names for the followingsubstances: (c) CH CH CH (a) C (b) CH2CNH2 CH2H2 CH (d) (e) (f) CHCH3 H H (g) (h) (i) n, :-SCH2CH3 3 CHH2CH2NHCH3 . OPO3- CH
-
If a particular glucose fermentation process is 87.0% efficient, how many grams of glucose would be required for the production of 51.0 g of ethyl alcohol (C 2 H 5 OH)? C 6 H 12 O 6 2C 2 H 5 OH +...
-
Suppose a small company does not want the headaches of administering benefits programs, so it hires its workers from a temp agency and keeps them on for several years. Would you expect the IRS to...
-
Propose structures for compounds E and F. Compound E (C8H6) reacts with 2 molar equivalents of bromine to form F (C8H6Br4). E has the IR spectrum shown in Fig. 9.50. What are the structures of E and...
-
The president of l&M Electric, a large regional utility, has asked that you perform a special engagement unrelated to the financial statement audit: Audit the customer payment system but with...
-
For the stamping department of a manufacturing firm, the standard cost for direct labor is $12 per hour, and the production standard calls for 1,000 stampings per hour. During June, 168 hours were...
-
The following chart is data over an 8-month period that shows how much a company spent in advertising and the sales revenue for that month MONTH ADVERTISING $ SALES $ March 900 56000 April 2700 89200...
-
Refer to the information pertaining to Endless Mountain Company that is provided in Integration Exercise 16. In addition to the budget schedules that you prepared in Integration Exercise 16, insert a...
-
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic ? system? N. Purine
-
A All-cis Cyclodecapentaene is a stable molecule that shows a single absorption in its 1H NMR spectrum at 5.67 ?. Tell whether it is aromatic, and explain its NMR spectrum.
-
What is the difference among corporate-level strategies, business-level strategies, and operating plans? How are they interconnected?
-
Calculate the change in accounts receivable based on the transactions below that took place in the same period: Cash sales: $650 Sales on credit: $780 Receipt from receivable: $420
-
The following data is from Chance Co.'s accounting records for year 2019: Units produced and sold Total revenues and costs Sales revenue Direct materials costs Direct labor costs Variable...
-
Anderson Corp. has the following information: Beginning Ending Inventory Inventory (1/1) (12/31) Direct Materials Inventory $20,000 $30,000 Work in Process Inventory $16,000 $18,000 Finished Goods...
-
The following is the adjusted trial balance of Sierra Company. Sierra Company Adjusted Trial Balance Account Title Cash December 31 Prepaid insurance Notes receivable (due in 5 years) Buildings...
-
A 2kg mass is moving at 10m/s along a frictionless surface towards a spring (at rest) with spring constant 50N/m. When the mass hits the spring, it will begin to compress it. What is the maximum...
-
X rays with a wavelength of \(0.20 \mathrm{~nm}\) undergo first-order diffraction from a crystal at a \(54^{\circ}\) angle of incidence. At what angle does first-order diffraction occur for...
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
What units should be used when expressing concentrations or partial pressures in the equilibrium constant? What are the units of K p and K c ? Explain.
-
Which configuration (R or S) does the bottom asymmetric carbon have for the D series of sugars? Which configuration for the L series?
-
(a) An aliphatic aminoglycoside is relatively stable to base, but it is quickly hydrolyzed by dilute acid. Propose a mechanism for the acid-catalyzed hydrolysis. (b) Ribonucleosides are not so easily...
-
All of the rings of the four heterocyclic bases are aromatic. This is more apparent when the polar resonance forms of the amide groups are drawn, as is done for thymine at the right. Redraw the...
-
What role do social institutions play in shaping individual and collective identities, and how do these identities intersect with race, gender, sexuality, and other aspects of social diversity to...
-
Blossom Ltd., which follows ASPE had the following comparative statement of financial position: Blossom Ltd. Comparative Statement of Financial Position December 31 Assets 2024 2023 Cash $84.000...
-
16. Let $V=\operatorname [span]\left\{\left(\begin{array}{1}1 \\ 0 \\ 1 \\ 0\end{array} ight), \left(\begin{array} (1)1 \\ 1 \\ 0 \\ 1\end{array} ight), \left(\begin{array} {1}0 \\ 1 \\ 1 AV...

Study smarter with the SolutionInn App


