Give the major product of each of the following reactions: a. b. c. d. e. f. g.
Question:
Give the major product of each of the following reactions:
a.
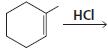
b.
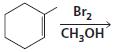
c.
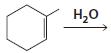
d.
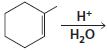
e.
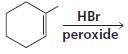
f.
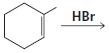
g.
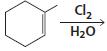
h.
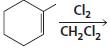
HCI CH3OH H20 Η, H+ H20 HBr peroxide HBr Cl2 H2O Cl2 CH2CI
Step by Step Answer:
a b c No rea...View the full answer



Related Video
Hydrogen peroxide can be used as a mild antiseptic to curb superficial skin infections such as athlete’s foot, but only in diluted quantities. To combat stinky feet, try soaking your feet in a solution of 1 part 3% hydrogen peroxide and 3 parts warm water for 15-20 minutes, then drying them thoroughly. This will kill odor-causing bacteria and soften your feet. To treat athlete\'s foot, you can use a similar solution, but only in diluted quantities, and soak your feet for 30 minutes. Hydrogen peroxide can also be used to keep vegetables fresh by adding 1/4 cup to a bowl of cold water, soaking the vegetables for 20-30 minutes, then draining, drying, and refrigerating them. Alternatively, you can spray vegetables with a solution of 3% hydrogen peroxide and let them stand for a few minutes before rinsing and drying. To keep leftover salad fresh, spray it with a solution of 1/2 cup water and 1 Tbsp. 3% hydrogen peroxide, drain, cover, and refrigerate.
Students also viewed these Organic Chemistry questions
-
Give the major product of each of the following reactions: a. b. c. d. e. f. HBr HC CCH3 peroxide excess Br 3 CH2Cl2 excesS CHC CCH HBr excess
-
Give the major product of each of the following reactions a. b. c. d. e. f. g. h. i. CCH3 + HNO3 S NO2 CHs CH CHCH NCH CH 3 CH3 HO CH3NCH3 + PC15- NC 1. H202 2. CH3 1. HO 3. Ht CH3 + CH:CH2MgBr 2.4...
-
Give the expected major product of each of the following reactions. PCC is the abbreviation for pyridinium chlorochromate (Section 8-6). (a) (b) (c) (d) (e) CH CH CH,OH NeCrO HSO, HO PCC, CH,CI (CH3)...
-
Formulate a plausible mechanism for the following reaction. The product is a precursor of mediquox (shown in the margin), an agent used to treat respiratory infections in chickens (no, we are not...
-
Highcountry.com is an Internet retailer of sporting good products. Customers order sporting goods from the company, using an online catalog. The company processes these orders and delivers the...
-
Why is inventory less liquid than accounts receivable?
-
\(\lambda_{1}\) and \(\lambda_{2}>0\left(\lambda_{1}, \lambda_{2} ight.\) : real and distinct \()\) a. Unstable node b. Saddle point c. Node d. Focus or spiral point e. Stable node
-
Presented below are data taken from the records of Alee Company. Additional information: 1. Held-to-maturity securities carried at a cost of $43,000 on December 31, 2013, were sold in 2014 for...
-
Bobby owns ten acres of land in rural Ohio, where he keeps a small herd of wild boar. Wild boar regularly attack humans. The attacks, which are usually unprovoked, can cause injury from bites and...
-
a. What does it mean when cash flow from operations on a company's cash flow statement is negative? Is this bad news? Is it dangerous? b. What does it mean when cash flow from investing activities on...
-
For each pair of bonds, which has the greater strength? Briefly explain why. a. b. c. d. CH,-Cl or CH,-Br CH,CH2CH2 or CH,CHCH3 CH, CH or CH, CH,CH, I-Br or BrBr
-
Using any alkene and any other reagents, how would you prepare the following compounds? a. b. c. d. e. f. CH3CH CH2CHCH CI CH2CHCH3 CH3CH2CHCHCH2CH3 Br CH CH2CHCHCH2CH3 Br Cl
-
Figure P1.13 depicts the various ways in which an average man gains and loses water in one day. One liter is ingested as food, and the body metabolically produces 0.3 liters. In breathing air, the...
-
True Or False A competitor is not privileged to puff if they know their statements are false and they intend to take business away from the plaintiff.
-
Under common law, defamation was a ____________ tort.
-
What is injurious falsehood? a. What is the difference between trade libel and slander of title? b. How does trade libel differ from defamation?
-
True Or False Public disclosure of private facts is a viable claim even if the details published are contained in a public record.
-
True Or False If an employer contracts for performance of an illegal act, they will be vicariously liable for damages caused by the contractor.
-
Spring Garden Vegetables (SGV) processes two brands of frozen vegetables, Valu-PAK and Gourmet. The two brands are processed in one factory using the same production process. The only difference...
-
Use the T account for Cash below to record the portion of each of the following transactions, if any that affect cash. How do these transactions affect the companys liquidity? Jan. 2 Provided...
-
Trans-1-Phenyl-1, 3-butadiene has max = 280 ( = 27,000) calculate the concentration of a solution that has A = 0.643 at 280nm in a 1 cm cell.
-
Nitro methane max = 275nm ( = 1.5) what kind of transition is responsible for this absorption?
-
3-Buten-2-one has max =213nm ( = 7080) and max = 320nm ( = 21) what kind of transition is responsible for each of these absorptions?
-
4 points Save Nwer Show what is produced by the following Java code. Assume the node's data fields are info and link, and the info is of type int. nodeA and nodeB are of reference variables of type...
-
Gyro Gear Company produces a single product, a special gear used in automatic transmissions. Each gear sells for $28, and the company sells 500,000 gears each year. Unit cost data are presented...
-
Cold water flows through a shower head steadily at a rate of 6 kg/min. The water is heated in an electric water heater from 25C to 45C. In an attempt to conserve energy,it is proposed to pass the...

Study smarter with the SolutionInn App


