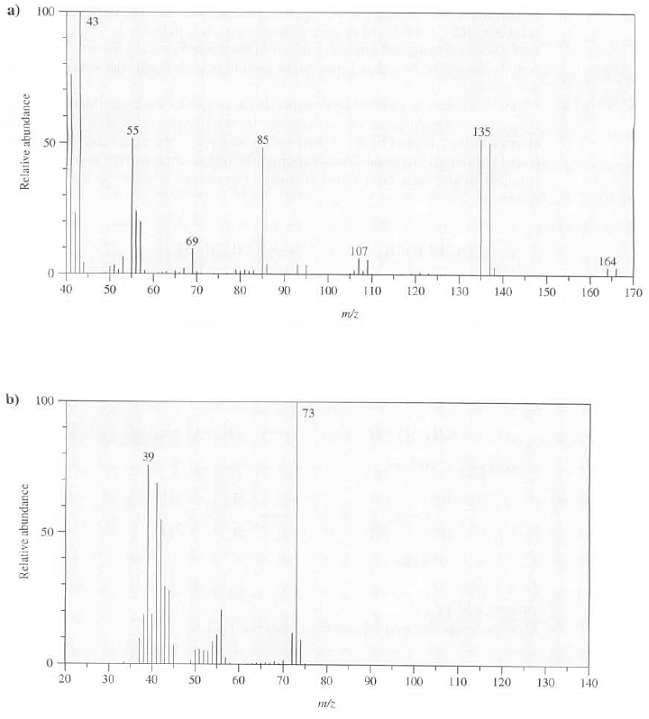
What conclusions can be drawn about these compounds from their massspectra? 100 43 55 135 85 69
Question:
What conclusions can be drawn about these compounds from their massspectra?
Transcribed Image Text:
100 43 55 135 85 69 107 164 trpt 40 50 60 70 80 L10 120 130 90 100 140 150 160 170 m/z b) 100 73 50 20 30 40 50 60 70 80 90 100 110 120 130 140 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a This similar intensities of the M mz 164 and th...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What conclusions can be drawn about income and health inequality between countries in the world? What is the general relationship between income and health status? Why do you think there is such a...
-
What conclusions can be drawn from a feasibility analysis?
-
What conclusions can be drawn from a study with a null result?
-
96. A 66-year-old woman with a long history of heavy smoking presents to her doctor with complaints of shortness of breath and chronic coughing that has been present for about 2 years and has been...
-
While performing interim audit procedures of accounts receivable, numerous unexpected errors are found resulting in a change of risk assessment. Which of the following audit responses would be most...
-
Rewrite the following paragraph to vary the length of the sentences and to shorten the paragraph so it looks more inviting to readers: Although major league baseball remains popular, more people are...
-
For each of the following situations, calculate the \(z\)-statistic \((z)\), make a decision about the null hypothesis (reject, do not reject), and indicate the level of significance \((p>.05, p
-
On January 1, 2014, Spalding Company sold 12% bonds having a maturity value of $1,000,000 for $1,075,814.74, which provides the bondholders with a 10% yield. The bonds are dated January 1, 2014, and...
-
1. As a Marketing Manager with responsibility for staff, describe three (3) issues that you see as most likely to create boundary-spanning problems for employees in a customer call center at your...
-
Tritium, or hydrogen-3, is prepared by bombarding lithium-6 with neutrons. A 0.250-mg sample of tritium decays at the rate of 8.94 1010 disintegrations per second. What is the decay constant (in /s)...
-
Predict the relative intensities of the M +, M + 2, and M + 4 peaks for these compounds. Assume that 79Br/81Br = 1/1 and 35Cl/37Cl=3/1. (a) CH2Cl2 (b) CH2BrCl
-
Show the molecular ions formed from these compounds: b) a) CH,NHCH,
-
List some arguments for and against the likelihood of convergence.
-
A proton, which moves perpendicular to a magnetic field of 1.4 T in a circular path of radius 0.074 m, has what speed?
-
Assume your AC thermostat is set so that the temperature in your home or apartment is maintained at 75 degrees F (24 degrees C) at all times during the summer. How much would you save each month if...
-
A marble rolls off a desk that is 0.86 m above the floor with a horizontal speed of 1.2 m/s. a. What is the marble's horizontal velocity when it is half-way to the floor? b. What is the marble's...
-
what is the heat produced in a conductor in 2 5 seconds if there is a current of 1 1 Amps and a resistance in the conductor of 7 . 2 ohms?
-
The cosmic microwave background has a peak spectral radiance frequency of 1 6 0 . 2 3 ghz . With this in mind, why was the planck telescope made to have a 1 . 5 m diameter mirror and to achieve a...
-
Which stage of group development is most closely associated with floundering and initial engagement? a. Forming b. Storming c. Norming d. Performing
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
In 1998, Robert Rubin, then the U.S. Treasury Secretary, claimed that the billions of dollars the U.S. government regularly provides to the IMF to make loans to other nations cost U.S. taxpayers not...
-
Write (+ and (- by the appropriate atoms and draw a dipole moment vector for any of the following molecules that are polar: (a) HF (b) IBr (c) Br2 (d) F2
-
Write bond-line formulas for (a) Four aldehydes and (b) Three ketones that have the formula C5H10O.
-
Write bond-line formulas for four carboxylic acids with the formula C5H10O2.
-
Why are gains/losses added back to (or deducted from) the cash from operations section of a cash flow statement?
-
Combination A, B, C, and F is the fifth most valuable with an NPV of $169,000. how did you came up with it?
-
Using a suitable example explain how you would check that equipment is ready for shut down to commence?

Study smarter with the SolutionInn App


