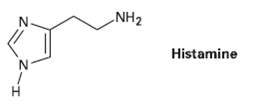
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List
Question:
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity, and explain yourordering.
Transcribed Image Text:
NH2 Histamine H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
b NH N bra CN Histamine 1 H The a nitrogen is mo...View the full answer

Answered By

Shikhar Srivastava
I have interest in science as it enables us to understand the process happening in our universe. I am a subject matter expert at Chegg solving advance physics problem with CF score more than 80% on average. I like to help student by solving problems. This helps me as well to build up my concepts regarding the subject. I have learnt from my experience that, In physics to build up command on subject one need to understand the theory, keep revising notes in free time and at last one more important thing is that one need to keep solving numerical problem related to the topic.
Taking career in core sciences is a long journey and one need to be calm minded with his or her profession.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain the basicity order of the following three amines: p-nitroaniline (A), rz-nitroaniline (A), and aniline (C). The structures and pKa data are shown in Table 23'l'
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
List the three states of matter in order of] (a) Increasing molecular disorder and (b) Increasing intermolecular attractions. (c) Which state of matter is most easily compressed?
-
Doug Brackett wants to have enough mechanics on hand to take care of his customer requests, but he does not want to be paying mechanics to sit around doing nothing. Doug needs to know a reasonable...
-
Based on the description in this case, how well would you say Susan Durbin appreciates the scope of human resource management? What, if any, additional skills of an HR professional would you...
-
Figure 20P-1 shows a hypothetical market for gasoline. a. Suppose an excise tax of $1.50 per gallon is levied on gasoline suppliers. Draw the after-tax supply curve. What price will consumers pay?...
-
Let the continuous random variable \(X\) denote the current measured in a thin copper wire in milliamperes. Assume that the range of \(X\) is \([4.9,5.1] \mathrm{mA}\), and assume that the...
-
The Village of Delmar is preparing its government- wide statement of activities for the year ended December 31, 2013. Analysis of the data accumulated thus far shows the following expenses for each...
-
Car A costs $10.37 per 100 miles to drive, while car B costs $6.66 per 100 miles to drive. How much more would car A cost to drive 550 miles? Car A costs $ more to drive 550 miles. (Type an integer...
-
In your opinion, based on the concepts presented within this chapter, what key fundamentals do managers need to understand to successfully manage a business?
-
Although pyrrole is a much weaker base than most other amines, it is a much stronger acid (pK a 15 for the pyrrole versus 35 for diethyl amine). The NH proton is readily abstracted by base to yield...
-
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain. Oxazole N:
-
Daisy Inc. issued $6 million of 10-year, 9% convertible bonds on June 1, 2023, at 98 plus accrued interest. The bonds were dated April 1, 2023, with interest payable April 1 and October 1. Bond...
-
What role do team-building rituals, traditions, and symbols play in shaping team identity and cohesion, and how can leaders leverage storytelling, ceremony, and shared experiences to reinforce...
-
How do leaders navigate the complexities of team dynamics, including conflicts, power struggles, and role ambiguity, while promoting constructive dialogue, conflict resolution, and mutual...
-
Letter Assignment For this assignment, I would like you to pretend that you are the coordinator for an upcoming trade show that will be held at the Columbus, OH Convention Center. You have received a...
-
Solve the differential equation with the given initial condition. 1x dx withx(10) = 2 dy 2 y 2y '
-
Recently, Syed, a director of an engineering firm, had to resolve a real workplace dispute that emerged when managers of the firm's two separate divisions (Geotechnical and Environmental) disputed...
-
Jamieson Enterprises was established recently. The balance of each item in its accounting equation is shown below for 6 March and for each of the following business days. Required: Assuming a single...
-
Explain what is meant by vicarious liability and when it is available?
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HNO3 b. HCI c. HBr d. HSO3
-
Using structural formulas, write equations for each of the following combustion reactions For information: (see Reaction Summary 1.a ): a. The complete combustion of propane b. The complete...
-
Write the formula for each of the following compounds: a. Isobutyl chloride b. isopropyl bromide c. 2-chlorobutane d. tert-butyl iodide e. Propyl fluoride f. general formula for an alkyl bromide
-
Using structural formulas, write equations for the following halogenation reactions (see Reaction Summary 1.b, p. 63), and name each organic product: a. The monochlorination of propane b. The...
-
Record the following transactions for Tri - State Corporation on the dates indicated. 1 . On March 3 1 , 2 0 2 0 , Tri - State Corporation discovered that Depreciation Expense on equipment for the...
-
William has decided to increase his sales price to $ 2 6 to offset the supplier's price increase. He believes that the increase will result in a 5 % reduction from last year's sales volume. What is...
-
The same scenarios exist as before, with labor costing either $ 1 3 / lawn or $ 2 5 / lawn . Assume that your mowing business will mow 3 5 0 lawns per month rather than 2 0 0 lawns per month. Also...

Study smarter with the SolutionInn App


