How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper
Question:
How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper reagent?
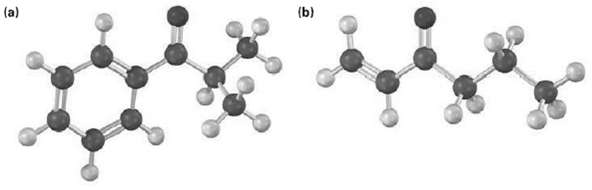
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Two combinations of acid chloride ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you prepare the following compounds with benzene as one of the starting materials? (a) (b)
-
How could you prepare the following amides using an acid chloride and an amine or ammonia? (a) CH3CH2CONHCH3 (b) N, N-Diethylbenzamide (c) Propanamide
-
a. Starting with isopropylacetylene, how could you prepare the following alcohols? 1. 2-methyl-2-pentanol 2. 4-methyl-2-pentanol b. In each case a second alcohol would also be obtained. What alcohol...
-
[10 marks] Click the link below to see the code for a program. Your task is as in assignment 3: re-write the code of this program so that, to a user of the program, it works as before, but in your...
-
Even with the popularity of online job-search sites, traditional job-search techniques are still important. What are some traditional sources for finding jobs?
-
Include correctly labeled diagrams, if useful or required, in explaining your answers. A correctly labeled diagram must have all axes and curves clearly labeled and must show directional changes. If...
-
Why is it necessary to create time-phased budgets in projects? What are their major strengths?
-
Suppose that technophiles are willing to pay $400 now for the latest iPhone, but only $300 if they have to wait a year. Normal people are willing to pay $250, and their desire to purchase does not...
-
This is a major ethical dilemma, in which AI systems may not be able to provide a truly neutral and fair system, thereby raising ethical concerns. If artificial intelligence tools are going to be...
-
Lakewood Tennis Club (LTC) operates an indoor tennis facility. The company charges a $ 150 annual membership fee plus a member rental rate of $ 20 per court per hour. LTCs fiscal year end is August...
-
Write the mechanism of the reaction just shown between 3, 4, 5-trimethoxybenzoyl chloride and morpholine to form trimetozine. Use curved arrows to show the electron flow in each step.
-
Write the mechanism of the reaction between p-hydroxyaniline and acetic anhydride to prepare acetaminophen.
-
Solve the following linear program graphically: Maximize cost = X1 + X2 8X1 + 16X2 64 X1 0 X2 2
-
Laker Incorporated s fiscal year - end is December 3 1 , 2 0 2 4 . The following is an adjusted trial balance as of December 3 1 . Accounts Debit Credit Cash $ 1 0 , 6 0 0 Supplies 3 2 , 0 0 0...
-
Sanders Company has the following information for last year: Selling price Variable production costs $190 per unit $52 per unit produced Variable selling and admin. expenses $18 per unit sold Fixed...
-
An investment opportunity requires an outlay of $x today but is not expected to have any cash inflows for the coming four years. At the end of year five its expected cash flow is $2,800 and that is...
-
A taxpayer needs help identifying and understanding their Adjusted Gross Income ( AGI ) for the tax year of 2 0 2 3 . Filing status: Single Their total income consists of the following: Wages $ 2 0 ,...
-
The following adjusted trial balance for Bell Servicing was prepared at the end of the fiscal year, December 3 1 , 2 0 2 3 3 1 , 2 0 2 3 : Account Debit Credit 1 0 1 1 0 1 Cash $ 7 , 3 0 0 7 , 3...
-
How does the hardness of a mineral relate to its chemical bonds?
-
What steps must a business take to implement a program of social responsibility?
-
According to MO theory, which molecule or ion has the highest bond order? Highest bond energy? Shortest bond length? 2- 02, 0, 0
-
Indicate whether the following peaks in the mass spectrum of 1-heptanol are odd-electron or even-electron ions. (a) m/z = 83 (b) m/z = 70 (c) m/z = 56 (d) m/z = 41
-
Calculate the energy in kJ mol-1 of the light described in Problem 12.1(b) Blue light with = 4800 A
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
Company: Anti spill cup company Competition analysis : Who are your competitors? Make a list of your potential competitors and rank them from least to most competitive. List the advantages your...
-
Henna Company produces and sells two products, Carvings and Mementos. It manufactures these products in separate factories and markets them through different channels. They have no shared costs. This...
-
Estimate the limit 29- lim t-2 -2x-4 x + 2x

Study smarter with the SolutionInn App


