How could you use Diels?Alder reactions to prepare the following products? Show the starting diene and dienophile
Question:
How could you use Diels?Alder reactions to prepare the following products? Show the starting diene and dienophile in each case.
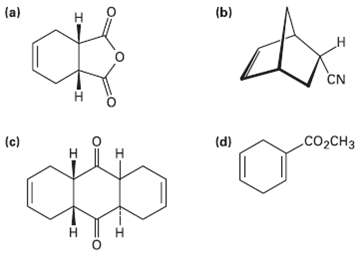
Transcribed Image Text:
(a) (b) н CN сооCнз (c) (d) н H. н т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
First find the cyclohexene ring formed by the DielsAlder ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you use 1H NMR spectroscopy to distinguish among the following esters? 018 18 - CH3 OH
-
How could you use expectancy theory to increase your own motivation level?
-
How could you use standard process costing to obtain information that helps improve the efficiency of the process?
-
An examiners close inspection of the annual financial statements and the accounting records revealed that Mawani Inc. may have violated some accounting principles. The examiner questioned the...
-
Discuss how well you think Starbuck's tuition reimbursement program meets the criteria for selecting employee benefits (organizational objectives, employees' expectations and values, and benefits...
-
Sulfur dioxide reacts with strontium oxide as follows: SO2(g) + SrO(s) SrSO3(s) (a) Without using thermochemical data, predict whether G for this reaction is more negative or less negative than H....
-
You are in the final stages of your audit of the Ozine Corporation's financial statements for the year ended December 31, 1999, when the corporation's chief executive officer, Gordon Sumner,...
-
The accounting records of Rhyme Redwood Corporation provide income statement data for 2012. Total revenue . . . . . . $ 940,000 Total expenses . . . . . . . 750,000 Income before tax . . . $ 190,000...
-
This is what the lab said to do Determine the circumference of the circle. For each trial, determine the period of the motion. For each trial, calculate the tangential velocity of the stopper. For...
-
Fantastique Bikes is a company that manufactures bikes in a monopolistically competitive market. The following graph shows Fantastique?s demand curve, marginal revenue curve (MR), marginal cost curve...
-
1, 3-Pentadiene is much more reactive in Diels?Alder reactions than 2, 4-pentadienal. Why might this be? H 1,3-Pentadiene 2,4-Pentadienal
-
Aldrin, a chlorinated insecticide now banned for use in the United States, can be made by Diels?Alder reaction of hexachloro-1, 3-cyclopentadiene with norbornadiene. What is the structure of aldrin?...
-
Can vertical analysis be used to compare two companies of different sizes, such as Walmart and Amazon.com, both dominant competitors in the retail industry? Explain.
-
Please answer to this discussion post below throughly and detailed. Police officers frequently face a moral dilemma that they may be tempted to believe a good end justifies bad means for what reasons...
-
Market solutions to environmental pollution fallaciously presume that what is good and rational for a collection of individuals is necessarily good and rational for a society. As a result, important...
-
What areas of law are pertinent to this case study ? Describe what the identified areas of law cover, and explain how they are relevant to the issues raised in the case study . (9 marks) (Question...
-
Write the expression as a single logarithm. 3 logm W logmz+5logmx 2
-
Big Waves is a large water park. Suppose the individual demand for entrance into Big Waves is Qd = 50 - (2 P) and each consumer has the same demand. Big Waves has a constant marginal cost of $5 per...
-
The generator initially provided \(125 \mathrm{~W}\) of power. If you assume that the power of the generator is proportional to the activity of the plutonium, by approximately what percent did the...
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
A solution contains a mixture of substance A and substance B, both of which are volatile. The mole fraction of substance A is 0.35. At 32 C the vapor pressure of pure A is 87 mmHg, and the vapor...
-
Propose a mechanism for the acid-catalyzed reaction of benzaldehyde with methanol to give benzaldehyde dimethyl acetal.
-
Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone dimethyl acetal.
-
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one (Figure 18-3)? In Figure 18.3 100 43 80 0 60 CH-C CH2CH 40 20 57 0 10 20 30 40 50 60 70 80 90 100 110 120...
-
Wendy runs a factory that makes stereo tuners. Each S100 takes 6 ounces of plastic and 4 ounces of metal. Each FS20 requires 3 ounces of plastic and 6 ounces of metal. The factory has 252 ounces of...
-
If the high temperature steam has a temperature of 2 9 7 . 7 K and the low temperature reservoir has a temperature of 1 4 7 . 8 K what is the carnot efficiency of this process
-
Discuss the innovative idea/project addressing the problem below by the following (a) potential impact - (b) novelty and innovation- (c) feasibility- (d) sustainability- Reference: i. Potential...

Study smarter with the SolutionInn App


