How many absorption are expected in the 1 H-NMR spectra of these compounds? CCH; .
Question:
How many absorption are expected in the 1 H-NMR spectra of these compounds?
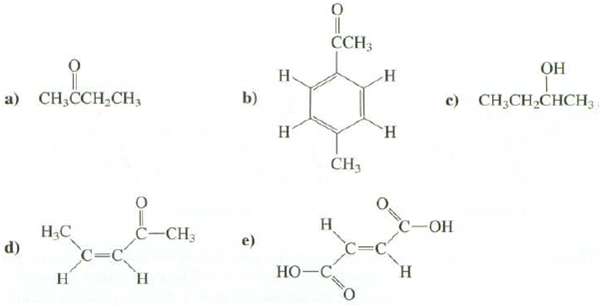
Transcribed Image Text:
CCH; ОН Н. н а) Cн,ССH,CH, c) CH;CH,CHCH3 b) н "н CH3 Н C=C н С -ОН Н.С d) С-СН, e) C= Н Но-С н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a The number of absorption signals in the NMR spectrum is equal to the number of different types of ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the 1H.NMR spectra of these compounds include the approximate chemical shift, multiplicity, and integral for each type ofhydrogen. CI b) CH;CHCH; ) C,CH,H c) CH,CH,OCH,CH3 CH2CH2NO2 f)...
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
NMR spectra for two compounds are given here, together with the molecular formulas. Each compound is a ketone or an aldehyde. In each case, show what characteristics of the spectrum imply the...
-
In determining an employee's net pay, which of the following taxes would be deducted? a. FUTA taxes b. SUTA taxes c. FICA taxes d. All of these choices are correct.
-
At mixing department, all materials are added at the beginning of the process. Lahore and overhead (conversion resources) are added evenly throughout the process. The following information pertains...
-
1. Are Connie and her staff on the right track to avoid manager mishaps by defining a new set of leader rules and core values and imposing it by fiat, from the top down? 2. Do you think a more...
-
For the HiTech example (17.1), find the risk-neutral probability of default. Example 17.1 (A leveraged firm) The HiTech firm currently has a value of $1 mil- lion and is financed in part by a 5-year...
-
Kitchen Electrics uses a perpetual inventory system. The following are three recent merchandising transactions: Apr. 10 Purchased 10 refrigerators from Polar Co. on account. Invoice price, $600 per...
-
Exactly two years ago, the U.S. Treasury issued 10-year Treasury notes at par value with a coupon rate of 2.50%. The Face Value of one of these notes was $10,000. Today, you want to purchase that...
-
Dalila Manufacturing Berhad (DMB) is suffering from the global economic effects for its main product. The product is sold in supermarkets throughout Malaysia. The following table shows the results of...
-
The absorption for the hydrogens of benzene appears 444Hz downfield from TMS on an instrument that operates of 60MHz. (a) Calculate the position of this absorption in units. (b) Calculate the...
-
Predict the approximate chemical shifts for the different hydrogen's in thesecompounds: CI CI a) CH,CH,CH3 b) CH;CHCH3 c) CH,COCH,CH3 d) CH;CHCH2
-
One of the following hydrocarbons is much more acidic than the others. Indicate which one, and explain why it is unusually acidic.
-
Calculate the present value PV of an investment that will be worth $1,000 at the stated interest rate after the stated amount of time. HINT [See Quick Example 4.] (Round your answer to the nearest...
-
In this experiment the accelerating voltage applied to the electron is V a = 1 . 9 6 0 = 1 . 9 6 0 x 1 0^3 1 0 Unknown node type: sup V . . The charge on an electron is e = 1 . 6 0 2 = 1 . 6 0 2 x 1...
-
Two spheres, one with charge Q-5uC and mass .010 kg, are attached 30 cm away from a second charge of q=-2uC, mass .005 kg. a. Label the forces acting on the spheres (draw the vectors) b. What is the...
-
Derik bought a promissory note this morning. Find the maturity value of the promissory note for $ 1 2 0 0 . 0 0 dated March 3 1 , 2 0 1 6 , and due on August 3 1 , 2 0 2 6 , if interest is 3 . 6 4 %...
-
During Module 6, you read about, heard lectures about, and thought about the relationship between employers and their employees. How should employers treat their employees? And vice versa. What do...
-
Prepare a group project for the classroom. Use as your model one of the following: a. Construction project b. Software development project c. Events management project (e.g., an awards banquet) d....
-
Refer to the data for problem 13-36 regarding Long Beach Pharmaceutical Company. Required: Compute each division's residual income for the year under each of the following assumptions about the...
-
Since the mid-1990s, individuals have been using online auction sites such as eBay, uBid, and Yahoo! to sell off old clothes, collectibles, and other used items that they find when cleaning out...
-
Propose a mechanism for the conversion of fumaroyl acetoacetate to fumarate plus acetoacetate (Problem 29.49).
-
Propose a mechanism for the conversion of acetoacetate to acetyl CoA (Problem 29.49).
-
Design your own degradative pathway. You know the rules (organic mechanisms), and you?ve seen the kinds of reactions that occur in the biological degradation of fats and carbohydrates into acetyl...
-
Solve the triangle shown to the right. Round the lengths of sides to the nearest tenth and angles to the nearest degree. 96 b=5 a=7 B A C C9.0 (Do not round until the final answer. Then round to the...
-
Hello, need help with making the program MealData.txt Avocado Egg Rolls 6.00 Parmesan-crusted Chicken 19.75 Strawberry Shortcake Pizookie 8.25 BBQ Tri-Tip Sliders 12.95 Spicy Peanut Chicken with Soba...
-
Assume you have been appointed to develop ethnic and multicultural marketing for a small chain of household appliance stores in large metropolitan area. There are several large concentrations of...

Study smarter with the SolutionInn App


