How many different absorption bands would appear in the 13C-NMR spectra of thesecompounds? b) CH;CH,CH,CH,CH3 c) CH
Question:
How many different absorption bands would appear in the 13C-NMR spectra of thesecompounds?
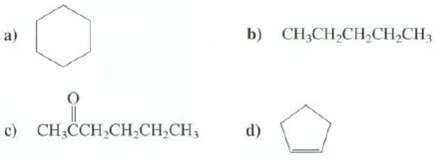
Transcribed Image Text:
b) CH;CH,CH,CH,CH3 c) CH CCH,CH;CH,CH, d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The number of absorptions in the 13CNMR spec...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The proton-decoupled 13C NMR spectra of 3-heptanol (A) and 4-heptanol (B) are given in Fig. 13.22 on page 626. Indicate which compound goes with each spectrum, and explain your reasoning. Fig. 13.22...
-
How many signals would you expect in the 13 C NMR spectrum of each of the compounds in Problem 16.34? In Problem 16.34 How many signals would you expect in the 1 H NMR spectrum of each of the...
-
The 1H and 13C NMR spectra of compound A, C8H9Br are shown. Propose a structure for A, and assign peaks in the spectra to your structure. TMS O ppm 10 8. 6. Chemical shift (8) TMS 200 180 160 140 120...
-
Is this a confined or unconfined aquifer? Please explain. 100 m amsl 78 m amsl 56 m amsl 48 m amsl A 50 m 11m B Clay Sand Clay 100 m
-
On January 1, 2011, Rand Corp. issued shares of its common stock to acquire all of the outstanding common stock of Spaulding Inc. Spaulding's book value was only $140,000 at the time, but Rand issued...
-
If you were a manager responsible for floor-cleaning products at a consumer products company, how might you apply crowd sourcing to identify a new product that would meet customer needs?
-
For each of the following sets of numbers, calculate a \(95 \%\) confidence interval for the mean ( \(\sigma\) not known); before going through the steps in calculating the confidence interval, the...
-
Based on records of automobile accidents in a recent year, the Department of Highway Safety and Motor Vehicles in Florida reported the counts who survived (S) and died (D), according to whether they...
-
Assume that CEDE Insurance Company has purchased from SS Reinsurance Company a surplus share treaty with a $5,000,000 limit and a retention of $70,000. CEDE has written three policies. Policy A...
-
Delaney AG leases an automobile with a fair value of 10,000 from Simon Motors, on the following terms. 1. Non-cancelable term of 50 months. 2. Rental of 200 per month (at the beginning of each...
-
Predict the 1H.NMR spectra of these compounds include the approximate chemical shift, multiplicity, and integral for each type ofhydrogen. CI b) CH;CHCH; ) C,CH,H c) CH,CH,OCH,CH3 CH2CH2NO2 f)...
-
Assign the absorptions in the 13C-NMR spectra of these compounds to the appropriate carbons: (a) 1-Butanol; absorptions at 61.4, 35.0, 19.1, and 13.6 (b) Cyclohexanone; absorptions at 209.7, 41.9,...
-
The shareholders' equity of Green Corporation includes $200,000 of $1 par common stock and $400,000 of 6% cumulative preferred stock. The board of directors of Green declared cash dividends of...
-
Man A (70kg) and Man B (90kg) are hanging motionless from a roof. If you cut the rope between Man A and Man B so that Man A stays motionless, what is the tension, TA, in the top rope? Assume the...
-
Item 18 Part A A capacitor consists of two square plates, 5.7 cm on a side, separated by a 2.0 mm air gap. How much energy would be stored in the capacitor if a mica dielectric is placed between the...
-
How much energy is stored by the electric field between two square plates, 8.4 cm on a side, separated by a 3.2 mm air gap? The charges on the plates are equal and opposite and of magnitude 330 C....
-
You are given two vectors A = -3.00 +5.00j and B=7.00 +2.00 Let counterclockwise angles be positive. What angle 84, where 0 < 0A <360, does make with the + z-axis? Express your answer in degrees. 0A...
-
A warehouse worker uses a forklift to lift a crate to a height 2.75 m above the floor. The combined mass of the platform and the crate is 207 kg. If the power expended by the forklift is 1440 W, how...
-
What is the lowest level of decomposition in the Work Breakdown Structure called? a. Work package b. Deliverable c. Subdeliverable d. Project
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
At FedEx, the marginal cost incurred in delivering packages speedily often encompasses more than just the expense of physically transporting a package from one location to another. The additional...
-
Have the following reactions taken place in a conrotatory or disrotatory manner? Under what conditions, thermal or photochemical, would you carry out eachreaction? (a) (b) --
-
What stereochemistryantarafacial or suprafacialwould you expect to observe in the following reactions? (a) A photo chemical [1, 5] sigma tropic rearrangement (b) A thermal [4 + 6] cyclo addition (c)...
-
The following thermal isomerization occurs tinder relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangementoccurs. CH C- CeH5. C&H5 "CH CD CoH5 CgH5 CEH5...
-
A project that is expected to generate annual after-tax cash flows of $2,200 for each of the next 20 years. The project's cost of capital is 8.5 percent and it has an IRR of 9.058 percent. What is...
-
Gabbie has to decide between the following two choices: Option 1: Get $320 with probability 35.97%, and $84.15 with the complement probability. Option 2: Get $94.95 with 100% probability. Her utility...
-
Why are unpaid accruals a "temporary" source of cash? Please give some details and examples.

Study smarter with the SolutionInn App


