How many protons, neutrons, and electrons are in each of the following atoms or ions? . 12
Question:
How many protons, neutrons, and electrons are in each of the following atoms or ions?
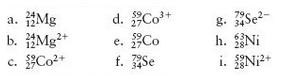
Transcribed Image Text:
. 12 e. 27 59NT:24
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a b c d e f ...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
How many valence electrons does each of the following atoms have? (a) Na (b) Cl (c) Si (d) B (e) Ne (f) N
-
How many significant figures are in each of the following numbers? a) 4.01 b) 4.010 c) 4 d) 2.00001 e) 0.00001 f) 2.1 - 1.10042 g) 7.01 3.1415
-
How many valence electrons do each of the following elements have, and what are the specific valence electrons for each element? a. Ca b. O c. element 117 d. In e. Ar f. Bi
-
58. HarperMotors sells 40 cars per month at an average price of $25,000. Thecarrying cost per car is $160 and the fixed order cost is $695. Howmany orders should the company place per year? 2 answers
-
What are Gardners original seven intelligences? Which two are the most closely associated with traditional IQ? What are the three new intelligences? Which of the 10 do you feel are most relevant and...
-
Consider the evaporator and control system in Figure 13.6. (a) Should each control valve be air-to-open (AO) or air-to-close (AC)? (b) Should each PI controller be direct-acting or reverse-acting?...
-
Should medical advice be dispensed on the telephone? Explain your opinion.
-
An inexperienced accountant for Nerys Corporation showed the following in the income statement: income before income taxes and extraordinary item $400,000, and extraordinary loss from flood (before...
-
Suppose an individual's utility function over income (M) and leisure (L) is U(M,L)-L, and the individual has non-labor income Y and earns wage w. The total amount of time available is 16 hours. a....
-
Barrett Chemicals manufactures four chemicals, Chem-1, Chem-2, Chem-3, and Chem-4, from a joint process. The total joint costs in May were $564,000. Additional information follows: Required Barrett...
-
Write the atomic symbol (A ZX) for each of the isotopes described below. a. Number of protons = 27, number of neutrons = 31 b. The isotope of boron with mass number 10 c. Z = 12, A = 23 d. Atomic...
-
Complete the following table. Number of Protons in Number of Neutrons in Number of Net Symbo Nucleus Nucleus Electrons Charge 238 92 20 23 20 28 20 89 39 35 15 36 16
-
Use data in Table 4H.1 or Appendix 2A to calculate the standard reaction entropy for each of the following reactions at 25C. For each reaction, interpret the sign and magnitude of the reaction...
-
What is meant by licensing?
-
What conclusions can be drawn from payback calculations?
-
What is the relationship between economic and met economic rationality?
-
What is the prerequisite for the use of profitability calculations?
-
Why do many business entities use a Balanced Scorecard?
-
What is three-layer design?
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
For the preceding question, which of the following equations best represents the reaction? a. 6N 2 + 6H 2 4NH 3 + 4N 2 b. N 2 + H 2 NH 3 c. N+ 3H NH 3 d. N 2 + 3H 2 2NH 3 e. 2N 2 + 6H 2 4NH 3...
-
The early alchemists used to do an experiment in which water was boiled for several days in a sealed glass container. Eventually, some solid residue would begin to appear in the bottom of the flask....
-
Name the following acids. A formula of Na2X. Which of the following statements is(are) true? Correct the false statements. a. The binary compound formed between X and fluorine will be a covalent...
-
Jet Airways borrowed $1,000,000 for one year to improve its liquidity. Jet Airways paid back the principal and interest of $1,060,000 at the end of the year. What is the dollar amount of interest and...
-
if the cost of attending the university is $40,000 per year due at the beginning instead of the end of each year for four years, how much do you have to deposit in a lump sum to be able to pay for...
-
Financial Analysis Case This case can be assigned as a group activity. Additional instructions and material for this activity can be found on the Instructor Resource site and in WileyPLUS. Kenmare...

Study smarter with the SolutionInn App


