How many types of nonequivalent protons are present in each of the followingmolecules? (c) (b) CH3CH2CH20CH3 (a)
Question:
How many types of nonequivalent protons are present in each of the followingmolecules?
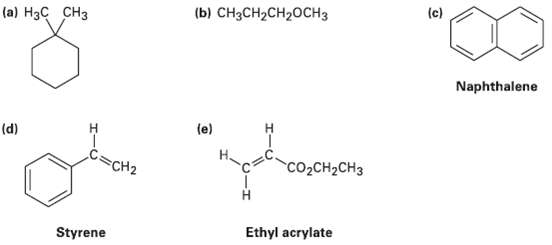
Transcribed Image Text:
(c) (b) CH3CH2CH20CH3 (a) H3C CH3 Naphthalene (e) (d) C=CH2 CO2CH2CH3 Ethyl acrylate Styrene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a 1 c Compound e 1 H3C CH3 2 3 2 pa 2 ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many asymmetric carbon atoms are present in each of the following compounds? (a) (b) (c) H-C-C-C-CI H CI H OH CH CH2OH OH H H OH
-
How many rings are present in each of the following compounds? Each consumes 2 moles of hydrogen on catalytic hydrogenation. (a) C8H8 (b) C8H8CI2 (c) C8H8O (d) C8H10O2 (e) C8H9ClO
-
How many rings are present in each of the following compounds? Each consumes 2 moles of hydrogen on catalytic hydrogenation. (a) C10H18 (d) C8H8O (b) C8H8 (e) C8H10O2 (c) C8H8CI2 (f) C8H9ClO
-
Under which of the following circumstances would an auditor be most likely to intensify an challenging examination of a $500 imprest petty cash fund a. Reimbursement occurs twice each week. b. The...
-
Most people rely on implicit costs in their everyday lives. Can you give some examples? What alternatives did you have to achieve the desire outcome?
-
A health-care researcher has obtained a sample of respondents from throughout the United States and has asked each one to indicate how worried he or she is about the avian flu on an interval-level...
-
Correlation between Residuals and Explanatory Variables. Consider a generic sequence of pairs of numbers \(\left(x_{1}, y_{1} ight), \ldots,\left(x_{n}, y_{n} ight)\) with the correlation coefficient...
-
You are a young CPA just starting your own practice in Hollywood, California, after five years experience with a Big 4 firm. You have several connections in the entertainment industry and hope to...
-
What would be the duration of the zero-coupon bond with 18 months to maturity, face value of $5,000,000, and a yield of 7.55% p.a.?
-
On October 1, 2020, Mary Graham organized a computer service company called Echo Systems. Echo is organized as a sole proprietorship and will provide consulting services, computer system...
-
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) C (c)
-
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (c) (b) (a) - HC- - H3C FCH2
-
Discuss potential investment opportunities for each of the following: a. An airline b. A tennis coaching clinic c. A café / Restaurant d. A fashion accessory company
-
What are the emergent properties of complex adaptive systems, and how do they manifest within ecological networks, influencing patterns of species coexistence, competition, and niche partitioning?
-
what ways do disturbances, such as natural disasters or anthropogenic activities, perturb the equilibrium of ecosystems, and what are the long-term implications for ecosystem structure and function?
-
Stacey Kessler has the following current-year passive income and loss amounts: $10,000 ABC ($18,000) XYZ What is the net income reported this year for the private interest activity if XYZ was...
-
A Johnson corporation bond is currently selling for $850. The bond matures in 20 years, has a face value of $1,000, and a yield to maturity of 10.55%. What is the bond's coupon rate ?
-
a. Describe any four (4) elements of fraud that are likely to be committed in a property insurance contract. (10 Marks) b. Explain why deductible is relevant to organizations such as UPSA and...
-
The weighted mean, denoted as \(\bar{x}_{w}\) is another measure of central tendency that can be used to assign weights (or measures of influence) to each of the individual observations in a sample....
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
Describe how to prepare each solution from the dry solute and the solvent. a. 1.00 * 10 2 mL of 0.500 M KCl b. 1.00 * 10 2 g of 0.500 m KCl c. 1.00 * 10 2 g of 5.0% KCl solution by mass
-
For each pair of compounds, describe which instrumental technique (IR, MS, proton NMR, carbon NMR) you could use to distinguish for certain which of the two compounds was in a sample. Describe what...
-
Hexamethylbenzene undergoes free-radical chlorination to give one monochlorinated product (C12H17Cl) and four dichlorinated products (C12H16Cl2). These products are easily separated by GC-MS, but the...
-
Show how you would distinguish among the following three isomers: (a) Using infrared spectroscopy and no other information. (b) Using proton NMR spectroscopy and no other information. (c) Using 13C...
-
Blue Company changed depreciation methods in 2025 from double-declining balance to straight-line. Depreciation prior to 2025 under double-declining-balance was 592,400, whereas straight-line...
-
Figure out, salaries, net income, add net income with retained earnings and totally stockholder equity , ?accounts payable and total liabilities and stockholders equity RILEY, INCORPORATED Statement...
-
Determine the material inventory balance at the end of may? Received Issued Receiving Received Materials Report Number Received Quantity Unit Price Requisition Number Issued Quantity Issued Balance...

Study smarter with the SolutionInn App


