How might you synthesize each of the following compounds using an aldol reaction? Show the structure of
Question:
How might you synthesize each of the following compounds using an aldol reaction? Show the structure of the starting aldehydes(s) or ketone(s) you would use in eachcase.
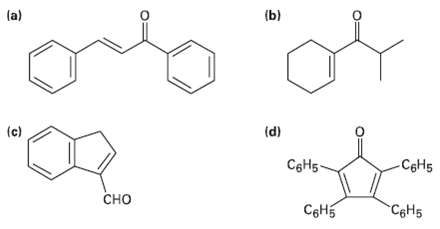
Transcribed Image Text:
(a) (b) (c) (d) CeH5 CeH5 - сно Cонь C&H5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
As always analyze the product for the carboncarbon double bond that is formed by ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you synthesize each of the following compounds from an acid chloride? (a) (b) ,-- NO, (CH),C-o-C-CH, C-o-c C CH2 C-O-C(CH)
-
Synthesize each of the following compounds starting from primary or secondary alcohols containing seven carbons or less and, if appropriate, bromobenzene. (a) (b) (c)
-
Show how you might synthesize each of the following compounds using, as your starting materials, esters, ketones, acyl halides, and so on: (a) (b) (c) OEt OEt
-
First United Bank Inc. is evaluating three capital investment projects by using the net present value method. Relevant data related to the projects are summarized as follows: Instructions 1. Assuming...
-
Many of the most pressing organizational issues attracting public attention seem to concern government agencies, especially those with responsibilities for preventing man-made disasters and attacks...
-
(a) What is meant by the term standard conditions with reference to enthalpy changes? (b) What is meant by the term enthalpy of formation? (c) What is meant by the term standard enthalpy of formation?
-
Give three examples of strategic technologies and how they can influence organizational performance.
-
Gainesville Cigar stocks Cuban cigars that have variable lead times because of the difficulty in importing the product: Lead time is normally distributed with an average of 6 weeks and a standard...
-
What are the implications of transformational leadership on employee motivation, organizational culture, and performance outcomes ?
-
On 1 January 2009 Henry Ltd issued a convertible debenture for 200 million carrying a coupon interest rate of 5%. The debenture is convertible at the option of the holders into 10 ordinary shares for...
-
Which of the following compounds would you expect to undergo aldol self-condensation? Show the product of each successful reaction. (a) Trimethylacetaldehyde (b) Cyclopentanone (c) Benzophenone...
-
What product would you except to obtain from aldol cyclization of hexane-dial, OHCCH 2 CH 2 CH 2 CH 2 CHO?
-
Use the graph to find the following. (a) Sign of the leading coefficient (b) Vertex (c) Axis of symmetry (d) Intervals where f is increasing and where f is decreasing 432 123 -y=f(x).
-
A VA medical center requires services for resurfacing the primary access road. They have stated the materials for the repair are petroleum-based and are concerned since oil prices are now at $4 a...
-
A French hotel operator which manages only hotels devoted to the MICE sector ( M eetings, I ncentives, C onventions, E xhibitions) let in August 2017 (term 2029) a 5-star hotel very close to...
-
According to the South African Rental Housing Act, 50 of 1990 (as amended by the Rental Housing Act 35 of 2014) answer the following questions below: Whose responsibility is it to promote rental...
-
The rate of homicides in Minneapolis MN, Chicago IL, How robberies affect victims The rate of date raping nationwide What are the most successful preventative approaches for DUIsearch design is...
-
Mr. Max Kassitta, married to Marta Kassitta with two children, had two Insurance policies, one a "life policy" with OMS Mutual (valued at N$ 850 000) and the other a "work accident insurance" with...
-
You hold a small ice cube near the top edge of a hemispherical bowl of radius \(100 \mathrm{~mm}\). When you release the cube from rest, what is the magnitude of its acceleration at the instant it...
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
Write the Lewis structure for each ion. Include resonance structures if necessary and assign formal charges to all atoms. If necessary, expand the octet on the central atom to lower formal charge. 3-...
-
The reaction of an amine with an alkyl halide gives an ammonium salt. The rate of this SN2 reaction is sensitive to the polarity of the solvent. Draw an energy diagram for this reaction in a nonpolar...
-
When water is shaken with hexane, the two liquids separate into two phases. Which compound is present in the top phase, and which is present in the bottom phase? When water is shaken with chloroform,...
-
aThe following reaction takes place under second-order conditions (strong nucleophile), yet the structure of the product shows rearrangement. Also, the rate of this reaction is several thousand times...
-
Consider a 15%, 20 year bond that pays interest annually, and its current price is $850. What is the promised yield to maturity?
-
You are a dual-income, no-kids family. You and your spouse havethe following debts (total): mortgage, $212,000; auto loan,$22,000; credit card balance, $16,000; other debts, $22,000.Further, you...
-
Can you please provide a feedback to this post? 1. What is the managerial view on business? What are the stakeholders view on business? How do they differ? The management viewpoint on business...

Study smarter with the SolutionInn App


