How would you carry out the following transformation? More than one step isneeded. CH3CH2CH2CH2C=CH T ? CCH2CH2CH2
Question:
How would you carry out the following transformation? More than one step isneeded.
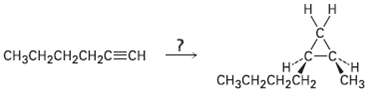
Transcribed Image Text:
нн CH3CH2CH2CH2C=CH Tн н? CнзCH2CH2CH2 СHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
Strategy The product contains a cisdisubstituted cyclopropane ring which can be formed from ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
How would you carry out the following reactions? More than one step may be required. (a) 3-Hexyne 3-Hexanone (b) Benzene m-Bromo acetophenone (c) Bromobenzene Acetophenone (d) 1-Methylcyclohexene ...
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Top executive officers of Preston Company, a merchandising firm, are preparing the next years budget. The controller has provided everyone with the current years projected income statement. Current...
-
Universal Technologies, Inc. has identified two qualified vendors with the capability to supply certain of its electronic components. For the coming year, Universal has estimated its volume...
-
The beam shown in the figure has a guided support at A and a spring support at B. The guided support permits vertical movement but no rotation. Derive the equation of the deflection curve and...
-
Yahoo is Fined $117.5 Million for Worst Data Hacks in History 1. Why do you think Yahoo was targeted for these data breaches? 2. Why did Yahoo keep the breaches from the public eye? How did their...
-
On January 1, the partners of Van, Bakel, and Cox (who share profits and losses in the ratio of 5:3:2, respectively) decide to liquidate their partnership. The trial balance at this date follows: The...
-
Identify some of the recommended actions that leaders can take to create conditions conducive to learning in an organization.
-
Air FranceKLM (AF), a Franco-Dutch company, prepares its financial statements according to International Financial Reporting Standards. AFs financial statements and disclosure notes for the year...
-
Each of the following syntheses requires more than one step. How would you carry themout? (a) CH3CH2CH2C=CH CH3CH2CH2CHO (b) (CH)2CHCH2CH CH-CH C=C (CH)2CHCH2 ?.
-
Synthesize the following compounds using 1-butyne as the only source of carbon, along with any inorganic reagents you need. More than one step may be needed. (a) 1, 1, 2, 2-Tetrachlorobutane (b) 1, 1...
-
Graph the annual CPI inflation rate and the annual GDP deflator inflation rate for Indonesia since 2000. What are the conceptual differences between these two measures of inflation? Judging from your...
-
What are the challenges of implementing Infrastructure as Code (IaC) in a large organization, and how do you address concerns related to configuration management and version control ?
-
How do you approach the problem of model interpretability in complex neural networks, and what tools or methods would you use to explain the decisions made by such models ?
-
3. Xander has an exclusive deal with Elite Spa and Casino to sell handbags. The average selling price is $450 per bag. Average variable costs (purchase price of bags plus freight costs and selling...
-
2. Why would investment companies go to the trouble of launching nanosatellites into space to improve their predictions? Why wouldn't they just rely on traditional sources of data to make these...
-
locate the Tesla balance sheet and determine through ratio analysis how effective the organization is at collecting receivables. How are the bad debt expense levels? Is there any cause for concern?...
-
A narrow span of control does not aid better communication between the superior and the subordinates. True False
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
A chemist wants to make 5.5 L of a 0.300 M CaCl 2 solution. What mass of CaCl 2 (in g) should the chemist use?
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
Draw the important resonance forms to show the delocalization of charges in the following ions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH-C CH H-C-CH CHCH CH2 NH CHCH- CH CH CH-CH 3 CH3 CH CH CH CH...
-
All of the following compounds can react as acids. Without using a table of acidities, rank them in order of increasing acidity. Explain your ranking. (a) CH3CH2SO3H (b) CH3CH2OH (c) CH3CH2COOH (d)...
-
As of June 30, Year 1, the bank statement showed an ending balance of $16,878. The unadjusted Cash account balance was $15,239. The following information is available: 1. Deposit in transit, $2,190....
-
Superior Company provided the following data for the year ended December 31 (all raw materials are used in production as direct materials): Selling expenses Purchases of raw materials Direct labor...
-
Marin Company produces two software products (Cloud-X and Cloud-Y) in two separate departments (A and B). These products are highly regarded network maintenance programs. Cloud-X is used for small...

Study smarter with the SolutionInn App


