How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required. .CH
Question:
How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required.
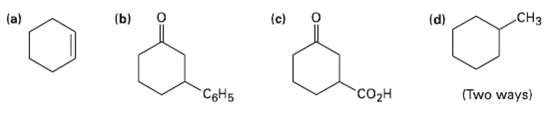
Transcribed Image Text:
.CHз (a) (b) (d) (c) сон CSH5 (Two ways)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
do HNNH KOH b d 1 LiC6H52 Cu ...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following substances from Cyclopentanol? More than one step may be required. (a) Cyclopentanone (b) Cyclopentane (c) 1-Methylcyclopentanol (d) Trans-2-Methylcyclopentanol I...
-
How would you prepare the following substances from 1-butanol? (a) Butyl amine (b) Dibutyl amine (c) Propyl amine (d) Pentylamine (e) N, N-Dimethyl butyl amine (f) Propene
-
How would you prepare the following substances from Pentanoic acid? (a) Pentanamide (b) Butyl amine (c) Pentylamine (d) 2-Bromopentanoic acid (e) Hexane nitrile (f) Hexylamine
-
Sherene Nili manages a company that produces wedding gowns. She produces both a custom product that is made to order and a standard product that is sold in bridal salons. Her accountant prepared the...
-
1. Review the first section of the chapter, about becoming world class. How can EatWell Technologies fulfill its social mission and be profitable? 2. In this case, where do you see resistance to...
-
A local newspaper headline blared, "Bo Smith Signed for $30 Million." A reading of the article revealed that on April 1, 2010, Bo Smith, the former record-breaking running back from Football...
-
A laminated \([0 / 90 / 0 / 90]_{s}\) carbon/epoxy beam is \(1 \mathrm{~mm}\) thick, \(20 \mathrm{~mm}\) wide, and has \(0.125-\mathrm{mm}\)-thick plies. The lamina properties are a. Determine the...
-
Coburn Furniture Company engaged in the following transactions during July of the current year: Jul. 2 Purchased inventory for cash, $12,800, less a quantity discount of $1,800. 5 Purchased store...
-
7.33 moles of a diatomic gas are at a temperature of 317 K. What is the internal energy of the gas?
-
Five jobs need to be done on a certain machine. However, the setup time for each job depends upon which job immediately preceded it, as shown by the following table: The objective is to schedule the...
-
Predict the products of the reaction of (i) phenyl acetaldehyde and (ii) acetophenone with the following reagents: (a) NaBH4 then H3O+ (b) Tollens reagent (c) NH2OH, HC1 catalyst (d) CH3MgBr, then...
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
Simkin's Hardware Store simulated an inventory ordering policy for Ace electric drills that involved an order quantity of 10 drills with a reorder point of 5. The first attempt to develop a...
-
IHRMs have unique challenges when it comes to managing an international assignment portfolio.1 Types of International Assignees and thoroughly address each of the following three items: Describe the...
-
How can scholars study ethical leadership when the subject is a term with different definitions?
-
A company invests a total of $30,000 of surplus funds at two annual rates of interest: 4% and 6 %. It wishes an annual yield of no less than 6 %. What is the least amount of money that the company...
-
Given your new credit score of 619, does your local banker have different incentives when it comes to evaluating your loan application? Explain.
-
Assume that a bond with $1000 par value and 7% coupon rate has a current price of $888. What is its current yield?
-
Conflicts of interest always reduce the flow of reliable information. Is this statement true, false, or uncertain? Explain your answer.
-
On October 1, 2021, Adoll Company acquired 2,600 shares of its $1 par value stock for $38 per share and held these shares in treasury. On March 1, 2023, Adoll resold all the treasury shares for $34...
-
Which of the following ionic compounds is composed of only nonmetal atoms? (a) NH 4 NO 3 ; (b) Al 2 (SO 4 ) 3 ; (c) Na 2 SO 3 ; (d) AlCl 3 ; (e) None of these.
-
When excess formaldehyde in basic solution is treated with ethanal, the following reaction takes place: Write a mechanism that accounts for the formation of the product.
-
When pseudoionone is treated with BF3 in acetic acid, ring closure takes place and α-and β-ionone are produced. This is the next step in the vitamin A synthesis. (a) Write...
-
(a) Write resonance structures for the anion of acetonitrile that account for its being much more acidic than ethane. (b) Give a step-by-step mechanism for the condensation of benzaldehyde with...
-
World Tourism Day 2 0 1 7 , with its official celebration on September 2 7 in Qatar, was focused on sustainable tourism. Established by the United Nations World Tourism Organization ( UNWTO ) , World...
-
Tracy is single and had adjusted gross income of $35,800 in 2018. Tracy also has the following items: How much may Tracy claim as itemized deductions?
-
Calculate the geometric mean return of an investment with five year returns of 10%, (15%), 12%, 8% and (5%).

Study smarter with the SolutionInn App


