How would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers?
Question:
How would you use NMR (either 13C or 1H) to distinguish between the following pairs of isomers?
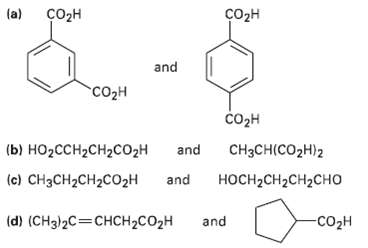
Transcribed Image Text:
(a) созн Cоон and со2н соон (b) НО2СCH2CH2CO2H CH3CH(CO2H)2 and (c) CH3CH2CH2C02H and носH-CH2CH2CHО and -со2н (d) (CH3)2C=CHCH2CO2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
In all of these pairs different numbers of peaks occur in the spectra of each iso...View the full answer

Answered By

Anoop V
I have five years of experience in teaching and I have National Eligibility in teaching (UGC-NET) .
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you distinguish between the following pairs of isomers by 1H NMR spectroscopy? a. CH3CCl3 and CH2ClCHCl2 b. CH3CH2CH2OH and (CH3)2CHOH c d. 0 C-OCH and H C OCH2CH CH2-CH=O and C CH
-
How would you use 1 H NMR spectroscopy to distinguish between the following compounds? (a) (b) (c) (d) (e) (f) . . . CI
-
Distinguish between the following pairs of terms: a. Debt securities classified as held to maturity versus available for sale b. Equity securities classified as trading securities versus available...
-
Write a critical review paper on the topic of financial management in the broad sense.
-
A wheelchair user is conducting a job search to land a middle-management position. Given that so many companies are attempting to build a more culturally diverse group of managers, should this job...
-
Juliana is taking out an $8,700, 3 1/2-year loan with an APR of 9.31%. What will be the monthly payment for this loan?
-
What are the relationships among Phases, Modes, and States of Operation?
-
The adjusted trial balance for Turnquist Company is given in E3-13. In E3-13, the trial balances before and after adjustment for Turnquist Company at the end of its fiscal year are presented below....
-
What are the key challenges and opportunities in cross-cultural negotiation, and how can negotiators leverage cultural differences to build trust and reach mutually beneficial agreements in diverse...
-
Pharoas Corporation paid $16,200 for a 90% interest in Sonista Corporation on January 1, 2019, when Sonista stockholders' equity consisted of $10,000 Capital Stock and $3,000 of Retained Earnings....
-
What spectroscopic method could you use to distinguish among the following three isomeric acids? Tell what characteristic features you would expect for eachacid. CH3(CH213CO2H (CH/2CHCH-cO2H...
-
Compound A, C 4 H 8 O 3 , has infrared absorptions at 1710 and 2500 to 3100 cm ?1 and has the 1 H NMR spectrum shown. Propose a structure for A. 11.18 TMS 10 O ppm 8. 6. 3. Chemical shift (6)...
-
Trade secrets do not have to be novel, be non-obvious, or have utility. If that is the case, then what is the economic advantage to a trade secret?
-
Sonya is selling her jet ski and has the following offers to choose fre 1. $3.500 today OR 2. $1.700 today and $1,900 in 5 months If money carns 6.3% simple interest a. What is the current economic...
-
Why is A/B testing so useful for marketing, and digital marketing in particular? Give at least two examples of A/B testing for marketing purposes. Give a null and alternative hypothesis for one of...
-
How sociocultural, economic, and political aspects influence economic development in Australia. With references please.
-
How do advancements in technology and virtual collaboration platforms reshape traditional paradigms of delegation in contemporary work environments ?
-
If a 15 cm diameter cast iron ball has a mass of 6.25 kg; then what diameter must a similar ball need in order to have a mass of 4.15 kg. Give your answer rounded to 3 significant digits.
-
A sinkhole is evidence of (a) wind erosion. (b) groundwater erosion. (c) ocean wave erosion. (d) stream erosion.
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
Molybdenum crystallizes with the body-centered unit cell. The radius of a molybdenum atom is 136 pm. Calculate the edge length of the unit cell and the density of molybdenum.
-
Draw a Lewis structure for acetonitrile, C2H3N, assuming that all bonding obeys the octet rule, and that no atom bears a formal charge. Acetonitrile contains a carbon-nitrogen triple bond.
-
Compute the formal charges on atom of the following structure. what is the charge on the entire structure? (a) :0: :0 P- :O:
-
Compute the formal charges on atom of the following structure. what is the charge on the entire structure? (a) :0: :0 P- :O:
-
2.1 Explain by means of drawings how the Successive-Approximation conversion process takes place when receiving an analog voltage input. Use the KEEP/RESET method. Use 5 V input. Vref= 8 V (10) 2.2...
-
Assume that we have a Ridge regression problem with only one predictor, and the true model is linear without an intercept, i.e. Y = X + e. Assume that we have In samples, (xi, y), (x2, Y2), . . .,...
-
Please write a program that calculates the final score of multiple students using different weight of exams and assign their final grade using the following criteria: If the average grade is 90 or...

Study smarter with the SolutionInn App


