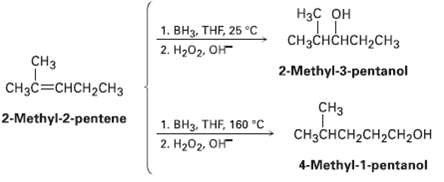
Hydroboration of 2-mnethyl-2-pentene at 25?C followed by oxidation with alkaline 11202 yields 2-methyl-3-pentanol, but hydroboration at 160?C
Question:
Hydroboration of 2-mnethyl-2-pentene at 25?C followed by oxidation with alkaline 11202 yields 2-methyl-3-pentanol, but hydroboration at 160?C followed by oxidation yields 4-methyl?1?pentanol. Suggest a mechanism.
Transcribed Image Text:
Нас он CHзснсHсH2сH3 1. ВНа, THF 25 °C 2. Hа02, Он CHз 2-Methyl-3-pentanol CH3C=CHCH2CH3 CH3 2-Methyl-2-pentene 1. BH3, THF, 160 "C CHзснсH2сH2CH2он 2. H202, OH 4-Methyl-1-pentanol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Hydroboration of 2methyl2pentene at 160C is reversible The ini...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Oxidation of an aldehyde yields a carboxylic acid: Draw the structures for the products of the following oxidation reactions. a. b. c. [ox] propanal 2,3-dimethylpentanal ox] 3-ethylbenzaldehyde>
-
Select any Company and get a copy of its annual report. Just write an outline of the company's annual report. The outline must contain the following: 1. Introduction 2. Background 3. Liquidity Ratios...
-
An abc-sequence balanced three-phase wye-connected source supplies power to a balanced wye-connected load. The line impedance per phase is 1 + j10, and the load impedance per phase is 20 + j20. If...
-
An ad for a cell phone service claims that its percent of "dropped calls" was significantly lower than that of its main competitor. In the fine print, the percents were given as 1.2 percent versus...
-
Suppose the initial condition is y(0) = 4. Find the solution with separation of variables and graph the result. What really happens at time t = 2? And what happens after this time? How does this...
-
Explain the concept of earned value.
-
Implementation of a new AIS went smoothly, for the most part. It is 15 years later, and now HMC is interested in mapping a variety of their business processes to determine whether improvements can be...
-
1. Research existing case law to analyze how the Supreme Court of the United States has ruled in an effort to achieve the Sixth Amendment's chief goal. a. Analyze at least four cases (three of which...
-
PDR plc manufactures four products using the same machinery. The following details relate to its products: (a) Determine the production plan which will maximize the weekly profit of PDR plc and...
-
Treatment of 4-penten-l-ol with aqueous Br2 yields a cyclic bromo ether rather than the expected bromohydrin. Suggest a mechanism, using curved arrows to show electronmovement. CH2B Br2, H20 %3...
-
We?ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions? 1 equiv Br2 CH3 (a) 2 equiv H2, Pd/C...
-
A velodrome is built for use in the Olympics. The radius of curvature of the surface is 20.0 m. At what angle should the surface be banked for cyclists moving at 18 m/s? (Choose an angle so that no...
-
A proton is moving in the uniform electric field of part A ( An electron is moving east in a uniform electric field of 1 . 4 5 N / C directed to the west ) . At point A , the velocity of the proton...
-
A 1 5 0 0 kg car is traveling on the road, to slow down at a rate 1 0 m / s how much force do the breaks need to apply to the car?
-
Determine the absolute pressure on the bottom of a swimming pool 3 0 . 0 m by 9 . 0 m whose uniform depth is 2 . 0 m . Determine the total force on the bottom of a swimming pool.
-
A very long thin wire carries a linear charge density of 2 . 3 3 x 1 0 - 5 C / m . Determine the electric field magnitude at a distance of 1 6 . 7 cm from the middle of the length of wire. ( K = 1 /...
-
Two stones are initially separated by a vertical distance y ( in meters ) from each other. One stone is thrown downward with ainitial velocity of - 1 7 m / s and the other is thrown upward from the...
-
The lithosphere includes the crust and the top part of the (a) asthenosphere. (b) mantle. (c) core. (d) lower mantle.
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
The results of a molecular orbital calculation for H 2 O are shown here. Examine each of the orbitals and classify them as bonding, antibonding, or nonbonding. Assign the correct number of electrons...
-
Show how the stereochemistry of the products will differ (if at all) when the following enantiomerically pure epoxide is hydrolyzed under acidic and basic conditions. D,C
-
(a) From what Grignard reagent can 3-methl-l pentanol be prepared by reaction with ethylene oxide, then aqueous acid? (b) Give the structure of another epoxide and another higher-order curpate that...
-
Explain why all attempts to isolate trimethyloxonium iodide lead instead to methl iodide and dimethl ether.
-
discuss the role of colonialism and its impact on language, identity, and power dynamics in Chinua Achebe's "Things Fall Apart," particularly in the context of the Igbo society and European...
-
Let Y1,...,Yn be a random sample from the following distributions with the unknown parameter(s). Estimate them by maximum likelihood and by the method of moments. fe (y) = 0y01, 0 y 1, 0 > 0
-
Julian and Samantha have resigned from their jobs in order to work together full-time on designing a software package that detects plagiarism in student assignments. They have already been able to...
Dimensional Analysis And Scale Up In Chemical Engineering 1st Edition - ISBN: 3540541020 - Free Book

Study smarter with the SolutionInn App


