Hydrocarbon A has the formula C 9 H 12 and absorbs 3 equivalents of H 2 to
Question:
Hydrocarbon A has the formula C9H12 and absorbs 3 equivalents of H2 to yield R, C9H18, when hydrogenated over a Pd/C catalyst. On treatment of A with aqueous H2SO4 in the presence of mercury (II), two isomeric ketones, C and D, are produced. Oxidation of A with KMnO4 gives a mixture of acetic acid (CH3CO2H) and the tricarboxylic acid E. Propose structures for compounds A?D, and write the reactions.
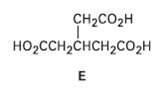
Transcribed Image Text:
CH2CO2H но-сCHснсH2Cо2н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
CHCHCH CH3 B A HOCCH ...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for compounds G-I: concd HNO heat 60-65 OH
-
Hydrocarbon A has the formula C12H8. It absorbs 8 equivalents of H2 on catalytic reduction over a palladium catalyst. On ozonolysis, only two products are formed: oxalic acid (HO2CCO2H) and succinic...
-
Propose structures for compounds E and F. Compound E (C8H6) reacts with 2 molar equivalents of bromine to form F (C8H6Br4). E has the IR spectrum shown in Fig. 9.50. What are the structures of E and...
-
Peabody Coal acquired the mineral rights to a tract of land containing coal deposits. Total costs of acquisition, exploration, development were $1,200,000 and an ARO of $300,000 was recorded. The...
-
During the next 8 months, Metropolis Power Company forecasts the demands shown in the table below (measured in thousands of kwh): Power will be supplied from the four generating facilities, GF1- GF4....
-
Consider the unusual piping diagram for the four tanks in Fig. E2.22 in which both the flow rates F 1 and F 2 are split between two streams entering the upper and lower tanks (denoted by the...
-
The owner of an ice cream store asks 75 people which flavor of ice cream they prefer. Thirteen of them say strawberry, 11 say chocolate, 24 say vanilla, and 27 provide a flavor other than strawberry,...
-
Peninsula Company owns an 80% controlling interest in Sandbar Company. Sandbar regularly sells merchandise to Peninsula, which then sells to outside parties. The gross profit on all such sales is...
-
Write a statement that assigns total_coins with the sum of nickel_count and dime_count. Sample output for 100 nickels and 200 dimes is: 300 Learn how our autograder works 542962 3875132.4x3zqy7 1...
-
Many municipalities are passing legislation that forbids smoking in restaurants and bars. Bar owners claim that these laws hurt their business. Are their concerns legitimate? The following data...
-
How would you carry out the following conversions? More than one step may be needed is some instances. CI RCH RCHCH3 RCH2CH3 RCH2CH2OH R-C=CH R-C C-CH3 RCH=CH2 R-C- RCCH3
-
How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d) CH . CH 7, 22H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHCH2CH2CH2C%CH (2 steps)
-
Digitex argues that once the objective of "achieving theoretical time" is clear, the challenge of achieving theoretical values becomes obvious and the necessary tools and approaches can be...
-
Costing Methods : Compare the job order and process costing methods, and explain how each of these can be applied to the company. How could the costs differ if one method is chosen over the other?...
-
Your employer agrees to give you a 10% raise after 1 year on the job, a 15% raise the next year, and a 20% raise the following year. Is your salary after the third year greater than, less than, or...
-
Sally Johnson desires to retire in 2 0 years. for vacation travel purposes, she wants to be able to withdraw $ 6 0 0 0 at the end of the first year of retirement, then increase withdrawals by $ 8 0 0...
-
Good discussion so far on the general application of the NPV and IRR formulas. Let's say the pizza oven is depreciated for 10 years, cost of the oven is $100,000, tax rate is 30%, it does not change...
-
Figure below shows a schematic phase diagram of Al - Cu alloy with Al melting point of 660 C, eutectic temperature 548 C, maximum solubility of Cu in Al = 5.65 wt%, and eutectic composition Xcu 33...
-
Suppose that the inverse demand function for movies is \(p=120-Q_{1}\) for college students and \(p=120-2 Q_{2}\) for other town residents. What is the town's total demand function...
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
To what volume should you dilute 25 mL of a 10.0 M H 2 SO 4 solution to obtain a 0.150 M H 2 SO 4 solution?
-
There is a small portion of the periodic table that you must know to do organic chemistry. Construct this part from memory, using the following steps. (a) From memory, make a list of the elements in...
-
For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and ionic. (a) NaCl (b) NaOH (c) CH3Li (d) CH2CI2 (e) NaOCH3 (f) HCO2Na (g) CF4
-
(a) Both PCI3 and PCI5 are stable compounds. Draw Lewis structures for these two compounds. (b) NCI3 is a known compound, but all attempts to synthesize NCI5 have failed. Draw Lewis structures for...
-
Evaluate the following limits. 3. lim f(x) if f(x) = 3 5x 2x, if x 1 4 (5 points) -3 1 + - 4x, if x < 1 x3 1 X - 1
-
Here are the height of 50 students who participated in a statistics survey: 242 23 34 60 45 66 77 23 34 45 674 54 52 54 65 76 89 89 76 56 76 34 43 33 47 56 58 266 16600 42 15 67 45 76 56 34 39 92 72...
-
Show that if f: [a, b] R is differentiable on (a, b) and f 0, then c = (a, b) such that f'(c) f(c) = + a-c 1 b-c Hint: Consider the function h(x) = f(x)(x - a)(x - b).

Study smarter with the SolutionInn App


