Identify each of the colored positions?red, blue, and green?as axial or equatorial. Then carry out a ring-flip,
Question:
Identify each of the colored positions?red, blue, and green?as axial or equatorial. Then carry out a ring-flip, and show the new positions occupied by each color,
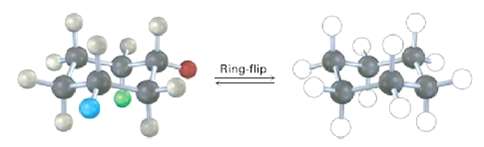
Transcribed Image Text:
Ring-flip
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
H I H I I H HB G equ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify each of the following as relating to (a) The control environment, (b) Control procedures, or (c) Monitoring. 1. Hiring of external auditors to review the adequacy of controls 2. Personnel...
-
Identify each of the following reactions as being a neutralization, precipitation, or oxidationreduction reaction. a. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g) b. Na2SO4(aq) + Hg(NO3)2(aq) HgSO4(s) +...
-
Identify each of the following elements as a metal, nonmetal, or metalloid: (a) Phosphorus (b) Strontium (c) Manganese, (d) Selenium (e) Sodium (f) Krypton.
-
Modify Lookup to make a program LookupAndPut that allows put operations to be specified on standard input. Use the convention that a plus sign indicates that the next two strings typed are the...
-
How do a closed-end fund, an open-end fund, and an exchange-trade fund differ?
-
Mandy and Jeff are parents of 2-year-old inhibited Sam and 3-year-old emotionally reactive Maria. Explain the importance of effortful control to Mandy and Jeff, and suggest ways they can strengthen...
-
Distinguish between a master file and a transaction file in an EDP system and indicate the counterpart of each in a manual system.
-
Coffee Bean, Inc. (CBI), is a processor and distributor of a variety of blends of coffee. The company buys coffee beans from around the world and roasts, blends, and packages them for resale. CBI...
-
Research a journal article on self-concept and self-esteem, summarize the article, and use that information along with your course readings to respond to the following. How can culture, ethnicity,...
-
Cranston Berries sells three types of berries: strawberries, blueberries, and raspberries. Sales have been booming this year and are expected to increase next year. The sales manager wants a program...
-
Draw two different chair conformations of trans-1, 4-dimethylcyclohexane, and label all positions as axial or equatorial.
-
What is the energy difference between the axial and equatorial conformations of cyclohexanol (hydroxycyclohexane)?
-
Calculate the power delivered to each resistor in the circuit shown in Figure P28.15. 2.00 3.00 1.00 18,0 V 4.00
-
What is the Equivalent Annual Rate (EAR) of a 12% Annual Percentage Rate (APR) compounded daily?
-
What terms or concepts come to mind when thinking about finance? Explain briefly
-
Which three factors out of the 5 ws and h are most important for business writing preparation?
-
How are bioremediation strategies leveraging microbial, enzymatic, and plant-based approaches to mitigate environmental pollution and promote sustainable remediation of contaminated sites ?
-
How does recombinant DNA technology facilitate the manipulation of genetic material to engineer novel organisms with desirable traits ? Explain
-
In early 2018, Qualcomm Inc. had \($15\) billion in debt, total equity capitalization of \($88\) billion, and an equity beta of 1.33 (as reported on Yahoo! Finance). Included in Qualcomms assets was...
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
A 50.0 g piece of iron at 152 C is dropped into 20.0 g H 2 O(l) at 89 C in an open, thermally insulatedcontainer. How much water would you expect to vaporize, assuming no water splashes out? The...
-
Starting with 2-methylpropene (isobutylene) and using any other needed reagents, outline a synthesis of each of the following (T = tritium, D = deuterium): (a) (b) (c) (d)
-
Show how you might carry out the following transformations: (a) (b) (c) (d) (e) OH Cl Ci OH
-
What compounds would you expect to be formed when each of the following ethers is refluxed with excess concentrated hydrobromic acid? (a) (b) (c) (d) (THF) (1,4-dioxane)
-
When you think of your existence in the world, how do you think of it? How do you exist in the world? What is the world? What are you? What is the relationship between you and the world? Now consider...
-
Requirement 2 Drop Down Options: 1. Generating ORUsing 2.Divesting itself of ORInvesting in new 3. Borrowing OR Issuing Stock.......Borrowing OR Issuing 4. Decreased OR Increased 5. Strong OR Weak...
-
Competitive Analysis/competitive landscape research was introduced this semester as an important quadrant of export market research. Using two case studies that we discussed only in class, illustrate...

Study smarter with the SolutionInn App


