Identify the functional groups in the following molecules, and show the polarity ofeach: (a) CH3CH2C=N (c) ()
Question:
Identify the functional groups in the following molecules, and show the polarity ofeach:
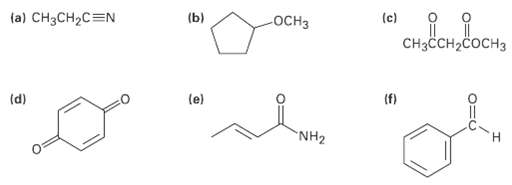
Transcribed Image Text:
(a) CH3CH2C=N (c) (ь) OCH3 CнзсCH-cоснз (f) (e) (d) "NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
a 8 d CH3CHCN nitrile d carboncarbon ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the functional groups in the following compounds. H H OH H HOCH,CCGCQ OH OH H OHH Glucose CHOC(CH CH, CHOC (CH CH 14 A triglyceride CH O A peptide H,C CH CH OH H,C H,C CHs Vitamin A
-
Identify the functional groups in the following model of arecoline, a veterinary drug used to control worms in animals. Convert the drawing into a line-bond structure and a molecular formula (red =...
-
Identify all of the functional groups in the following molecule. OH HOO
-
Label the following as True, False, or Uncertain and explain your choice. (Uncertain means that it can be either true or false depending upon the circumstances.) a. All members of a resource cartel...
-
1. How would you rate the financial status of the Garners before the air conditioner broke down? 2. The Garners take-home pay is over $4,500 a month. Yet, after all expenses are paid, there is only...
-
The compression ratio of an air standard Otto cycle is 8.7. Prior to the isentropic compression process, air is at 120 kPa, 19oC, and 660 cm3. The temperature at the end of the isentropic expansion...
-
Write a program that solves either a) the Towers of Hanoi problem with up to 1000 disks, or, b) the Traveling Salesman problem with up to 10 cities. You may need to wait until you have read about...
-
The town barber shop can accommodate 35 customers per day. The manager has determined that if two additional barbers are hired, the shop can accommodate 80 customers per day. What are the design and...
-
Cricket's Indian Premier League (IPL) uses an English auction to assign players to teams each year. In an English auction, the teams publicly announce their successive higher bids until no higher bid...
-
In December 2018, the Accounts Receivable controlling account on the books of Stag Co. showed one debit posting and two credit postings. The debit represents receivables from December saIes of...
-
Look at the following energy diagram for an enzyme-catalyzed reaction: (a) How many steps are involved? (b) Which step is most exergonic? (c) Which step is the slowest? Energy
-
Identify the following reactions as additions, eliminations, substitutions, orrearrangements: (a) CH3CH2Br + NACN CH3CH2CN (+ NaBr) (b) Acid (+ H20) OH cataiyst (c) Heat NO2 "O. (d) Light (+ HNO2)...
-
Guehler Electric sold $2,000,000, 9%, 10-year bonds on January 1, 2014. The bonds were dated January 1 and pay interest July 1 and January 1. Guehler Electric uses the straight-line method to...
-
It has been found that Angstrom Incorporated's thermal control system has failed due to a mercury-filled bulb sensor failure. Modern regulations prohibit mercury sensors from being used in thermal...
-
a solid culindral is subjected to torque of 2 8 6 4 7 . 8 Nm 1 . if the shear stress is not to exceed 8 0 N / mm ? Find it diameter 2 . What percent saving in weight would be obtained if this shaft...
-
Estimate how much collector area and storage capacity would be required for an active solar hot water system designed to supply the total needs for two four-person families, one living in Manchester,...
-
An arc-welding operation on nickel performs a groove weld, whose cross-sectional area = 29 mm 2 . Travel velocity = 3.9 mm/s. Heat transfer factor = 0.83 and melting factor = 0.64. Determine the rate...
-
The voltage in an EBW operation is 39 kV. The beam current is 39 milliamp. The electron beam is focused on a circular area that is 0.5 mm in diameter. The heat transfer factor is 0.87. Calculate the...
-
Tsunami (a) are usually caused by hurricanes. (b) are usually caused by global warming. (c) are usually caused by the Milankovitch cycles. (d) are usually caused by earthquakes.
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
The hybridization scheme for the central atom includes a d orbital contribution in (a) I 3 - ; (b) PCl 3 ; (c) NO 3 - ; (d) H 2 Se.
-
What monobromination product (or products) would you expect to obtain when the following compounds undergo ring Bromination with Br2 and FeBr3? (a) (b) (c) N.
-
Predict the major products of the following reactions: (a) (c) (e) Product of (c) + H2O Product of (c) + H2 (1 molar equivalent) (g) Product of (f) HCI Styrene EtONa HA, heat OH peroxides HA heat Pt...
-
Starting with benzene, outline a synthesis of each of the following: (a) Isopropylbenzene (b) tert-Butylbenzene (c) Propylbenzene (d) Butylbenzene (e) 1-tert-Butyl-4-chlorobenzene (f)...
-
With a one day sale on Christmas Day and same day return policy in full effect, people filled the stores of Macy's to shop and return. The total revenue sales numbers were 2M with $150,000 in...
-
An ethical organizational culture creates an environment in which to structure behavior that is then evaluated by stakeholders. The key elements of an organizational culture include all of the...
-
Identify and discuss the seven (7) key dimensions of organizational culture Describe what ZenDesk's organizational culture looks like Discuss how ZenDesk uses its corporate culture to attain...

Study smarter with the SolutionInn App


