If methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather
Question:
If methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather than an acid is produced. Show how you could carry out the following transformation, and propose a mechanism for the ester-formingstep.
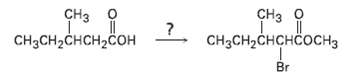
Transcribed Image Text:
CHз о CHз о сHзCH-CHCHCOСH3 CH3CH-CHCH-COн Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
OH H CHCH3CHCH3 0 PBr3 Br formation of acid bromide HCH3 H Br Br H CHCH3CHCH3 enoliz...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How much mass should be at the end of a spring (k = 100 N/m) in order to have a period of 2.0 s?
-
Show how you could prepare each of the following compounds. Use the starting material indicated along with ethyl acetoacetate or diethyl malonate and any necessary inorganic reagents. Assume also...
-
The gate at the end of a 1-m-wide freshwater channel is fabricated from three 125-kg, rectangular steel plates. The gate is hinged at A and rests against a frictionless support at D. Knowing that...
-
Determine the maximum weight of the crate so that the tension developed in any cable does not exceed 450lb. 2 ft 1ft 2 f 2 ft,
-
Why are reports indispensable documents in business?
-
Graph each function in problem over the indicated interval. 200 0,51 ] N = 1 + 3e-r [
-
Explain the difference between the STRIPS language and the ADL language. Why is ADL described as being more expressive than STRIPS? What kinds of problems might ADL be used to solve for which STRIPS...
-
Ten mL of pure liquid water in a cylinder with a movable piston is heated at a constant pressure of 1 atm from an initial temperature of 80?C. The temperature of the system is monitored, and the...
-
Nittany Company uses a periodic inventory system. At the end of the annual accounting period, December 31 of the current year, the accounting records provided the following information for product 1:...
-
Bug-Off Exterminators provides pest control services and sells extermination products manufactured by other companies. The following six-column table contains the company's unadjusted trial balance...
-
Show how you might prepare 1-penten-3-one from 3-pentanone.
-
Identify the most acidic hydrogens in each of the following molecules: (a) CH3CH2CHO (b) (CH3) CH3CCOCH3 (c) CH3CO2H (d) Benz amide (e) CH3CH2CH2CN (f) CH3CON (CH3)2
-
a. You expect an RFR of 10 percent and the market return (RM) of 14 percent. Compute the expected return for the following stocks, and plot them on an SML graph. b. You ask a stockbroker what the...
-
Company XYZ has a current stock price of $39.43 and is not expected to pay dividends in years 1, 2 and 3. it is expected to pay a dividend of S8 four years from today. After year 4, dividends are...
-
You want to be a millionaire when you retire in 30 years. You CANNOT put any money away for the next 10 years, but will start the month after that. How much do you have to save each month if you can...
-
The current spot exchange rate is RM5.76/ and the three-month forward rate is RM5.70/. Based on your analysis of the exchange rate, you are pretty confident that the spot exchange rate will be...
-
The firm originally is 100% financed by equity ( Unlevered firm ). The EBIT for the firm is $20,000. The cost of capital for this unlevered firm is 10%. The tax rate is 40%. Systematic risk of the...
-
You have been asked to evaluate the proposed acquisition of a new Machine for your company. The machine's basic price is $150,000. Assume that the machine can be depreciated using straight line over...
-
An igneous rock can be transformed into a metamorphic rock by (a) weathering, erosion, deposition, then compaction to become a sedimentary rock, then heat and pressure. (b) being squeezed by great...
-
The following T-accounts show postings of selected transactions. Indicate the journal used in recording each of these postings a through e. Cash Accounts Receivable Inventory (d) 500 (e) 300 (b)...
-
How is the number of hybrid orbitals related to the number of standard atomic orbitals that are hybridized?
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
Would l-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at carbon-5 Explain your reasoning.
-
For the following exercises, solve each system by Gaussian elimination. 17. 2x-y+32 17 -5x+4y-22=-46 2y+5x=-7 20. 4x+6y 2z = 8 6x+9y-32-12 -2x-3y+2=-4 23. x+y+2=14 2y+32=-14 -16y-242 -112...
-
Wildhorse Manufacturing incurs $82,900 of direct labour costs. Supporting records show that the assembly department used $47,500 of the direct labour and the finishing department used the remainder....
-
Explain and contrast unique ( individual , unsystematic ) risk of a firm and market ( systematic ) risk. Explain how unique risk and market risk can be measured.

Study smarter with the SolutionInn App


