In Figure R 1 = R 2 = 10.0?, and the ideal battery has emf ? =
Question:
In Figure R1 = R2 = 10.0?, and the ideal battery has emf ? = 12.0 V.
(a) What value of R3 maximizes the rate at which the battery supplies energy and
(b) What is that maximum rate?
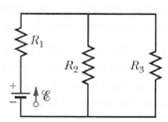
Transcribed Image Text:
Rs R2 ww ww
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
With the unit understood the equivalent resistance for ...View the full answer

Answered By

Ishita Barary
I have completed my B.Sc Honours in Botany from Scottish church College under University if Calcutta with high first class marks in 2014. Then I completed my M.Sc degree from University of Calcutta in Botany with specialization in Molecular Biology and Biotechnology with high first class marks in 2016.I have completed my M.Sc project in Bioinformatics.Currently I am doing Ph.D in Botany from Visva-Bharati University(A Central University).I have teaching experience of 5 years in B.Sc honours as a private tutor.I am providing GATE tuition as I am GATE qualified.I also provide 11 & 12 Biology tuition and I have more than 2 years of experience on that.I have one paper on my M.sc project in UGC listed journal.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
In Figure the inductor has 25 turns and the ideal battery has an emf of 16 V. Figure gives the magnetic flux through each turn versus the current i through the inductor. The vertical axis scale is...
-
In Figure, R = 15??, L = 5.0 H, the ideal battery has = 10 V and the fuse in the upper branch is an ideal 3.0 A fuse. It has zero resistance as long as the current through it remains less than 3.0...
-
Figure R 1 = 6.00?, R 2 = 18.0? and the ideal battery has emf ? = 12.0 V. What are the (a) Size and (b) Direction (left or right) of current it? (c) How much energy is dissipated by all four...
-
(a) A proton is moving at a speed much slower than the speed of light. It has kinetic energy K1 and momentum P1. If the momentum of the proton is doubled, so P2 = 2p1 how is its new kinetic energy K2...
-
Northern Communications has the following stockholders' equity on December 31, 2018: Requirements 1. Assuming the preferred stock is cumulative, compute the amount of dividends to preferred...
-
Hatfield and McCoy hate each other but love corn whiskey. Because they hate for each other to be happy, each wants the other to have less whiskey. Hatfields utility function is UH(WH,WM) = WHW2M and...
-
Fill in the Blank. Real-time analyzers are useful for machinery ___________ monitoring, since a change in the noise or vibration spectrum can be observed immediately.
-
During the last week of August, Oneida Companys owner approaches the bank for an $ 100,000 loan to be made on September 2 and repaid on November 30 with annual interest of 12%, for an interest cost...
-
Based on the following data, how much of a refund would Rip and Beth receive after filing their income tax return? ( Input the amount as a positive value. Round your intermediate calculations and...
-
How many solutions does the equation ||2x 3 m| = m have if m > 0? - A 1 2 D 3. 4- B
-
In Figure R 1 = 10.0?, R 2 = 20.0? and the ideal batteries have emf s ? 1 = 20.0 V and ? 2? = 50.0 V. What value of later, results in no current through battery 1? ww R. Rg
-
Each of the six real batteries in Figure has an emf of 20 V and a resistance of 4.0. (a) What is the current through the (external) resistance R = 4.0? (b) What is the potential difference across...
-
Why is the question of up or down milling more critical in horizontal slab milling than in vertical spindle (end or face) milling?
-
Technology has opened many doors. Just one of those doors for businesses large or small is the ability to launch and sustain a presence in the global market. However, this is easier said than done....
-
A large company is planning to replace its application for employees time reporting with a new application. The seller of the new application indicated that 9 0 % of its customers are happy with...
-
1. When developing a financial plan for a corporation you should consider which of the following? I. How much net working capital will be needed? II. Will additional fixed assets be required? III....
-
Slick Mountain Ski Rental Company incurred the following costs and ski rental hours during the following three months of the current year. Assume that the driver for all variable costs is ski rental...
-
Use the decomposition ALU to solve the system Az = b, where 1 0 4 1 L= 3 1 0U=0 -2 2 b= 24 1 31 0 0 3 First solve Lyb, and then solve Uz = y.
-
At what minimal radius along the perpendicular bisector of a wire of length \(0.25 \mathrm{~m}\) carrying \(30 \mathrm{nC}\) of charge does the assumption of cylindrical symmetry in applying Gauss's...
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
Find the derivatives of (g(x)) and g((u)). (u) = u 3 , g(x) = 1/x + 1
-
Cell Membranes Cell membranes (the walled enclosure around a cell) are typically about 7.5 nm thick. They are partially permeable to allow charged material to pass in and out, as needed. Equal but...
-
Electronic flash units for cameras contain a capacitor for storing the energy used to produce the flash. In one such unit, the flash lasts for 675S with an average light power output of 2.70 X 105W....
-
In one type of computer keyboard, each key holds a small metal plate that serves as one plate of a parallel-plate, air-filled capacitor. When the key is depressed, the plate separation decreases and...
-
Townsend Produce agrees to sell 100 crates of lettuce to Metro Grocers under a shipment contract. Townsend arranges for Excel Transport to pick up and deliver the lettuce, which Excel Transport does....
-
Assume an organic compound has a partition coefficient between water and ethyl acetate equal to 8.12. If there are initially 7.10 grams of the compound dissolved in 75.0 mL of water, how many grams...
-
NO. DATE Use the table below Write a Java program that will display the output below. code may vary... For your reference QUANTITY ITEM PRICE 1 Bond Paper (rim) 230 1 Notebook 30 1 1 Printer Ink 5500...

Study smarter with the SolutionInn App


