In light of the fact that tertiary alkyl halides undergo spontaneous dissociation to yield a carbocation plus
Question:
In light of the fact that tertiary alkyl halides undergo spontaneous dissociation to yield a carbocation plus halide ion, propose a mechanism for the followingreaction:
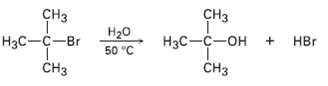
Transcribed Image Text:
CHз сHз Нзс—с—он + НBr CHз Нзс —с—Br Нао 50 "C CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
CH3 HCCBr CH...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In view of the fact that hot air rises, why does it generally become cooler as you climb a mountain? (Note that air has low thermal conductivity.)
-
In view of the fact that hot air rises, why does it generally become cooler as you climb a mountain? (Note that air has low thermal conductivity.)
-
In Example 26.2, Janets handling of the fact that her data showed 26 volunteers in the control group when there should have been only 25.
-
The balance sheet data for Alans Lightworks, Corp., at August 31, 2012, and September 30, 2012, follow: Requirement 1. The following are three independent assumptions about the business during...
-
Sketch graphs for the relationships described in each of the following problems and select one or more of the families of functions discussed in Section 2.4.4 to represent it. 1. The relationship...
-
Write balanced chemical equations to correspond to each of the following descriptions: (a) When sulfur trioxide gas reacts with water, a solution of sulfuric acid forms. (b) Boron sulfide, B2S3(s),...
-
Explain the differences between the various types of e-commerce marketplaces.
-
After analyzing the costs of various options for obtaining brackets, Ross White (see Problems 6-25, 6-26, and 6-27) recognizes that although he knows that lead time is 2 days and demand per day...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date January 1 January 10 March 14 March 15 July 30. October 5 Activities Beginning inventory Sales...
-
A random sample of likely voters showed that 49% planned to support Measure X. The margin of error is 3 percentage points with a 95% confidence level. a. Using a carefully worded sentence, report the...
-
Tertiary alkyl halides, R 3 CX, undergo spontaneous dissociation to yield a carbocation, R 3 C + , plus halide ion. Which do you think reacts faster, (CH 3 ) 3 CBr or H 2 C = CHC (CH 3 ) 2 Br?...
-
Carboxylic acids (RCO 2 H; p K a 5) are approximately 10 11 times more acidic than alcohols (ROH; pK a 16). In other words, a carboxylate ion (RCO 2 ) is more stable than an alkoxide ion (RO )....
-
Yount Company reports the following for the month of June. Instructions(a) Calculate the cost of the ending inventory and the cost of goods sold for each cost flow assumption, using a perpetual...
-
https://youtu.be/rlBk0CH1rYY Review the budget in the document on the next slide. Using your knowledge that you have gained so far, answer the questions in your journal. Provide as much detail as...
-
Moody Corporation uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company made the following estimates:...
-
On July 1, 2023, Ivanhoe Inc. issued $330000, 9% bonds, which mature on July 1, 2030. The bonds were issued for $313934 to yield 10%. Ivanhoe uses the effective interest method of amortizing bond...
-
The combinor paid $480 to acquire 75% of common shares issued by combinee on 1st January 2022. The fair market value of net assets of combinee 400. What is the goodwill of the subsidiary on this date?
-
Question 5 (25 marks) Siyabakulisa (Pty) Ltd is a small manufacturing company. The company's accounting functions are carried out by the accounting staff consisting of the accountant, Zandile Zulu,...
-
The Foodpanda group is a worldwide mobile food delivery marketplace headquartered in Berlin, Germany. It was launched in March 2012. It allows you to select from local restaurants and place orders...
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
Draw the Lewis structure for urea, H 2 NCONH 2 , one of the compounds responsible for the smell of urine. (The central carbon atom is bonded to both nitrogen atoms and to the oxygen atom.) Does urea...
-
Which of the following pairs of compounds could be separated by recrystallization or distillation? (a) meso-tartaric acid and (±)-tartaric acid (HOOC-CHOH-CHOH-COOH) (b) (c) (d) CH,CH, O CH,CH...
-
To show that (R)-2-butyl (R, R)-tartrate and (S)-2-butyl (R,R)-tartrate are not enantiomers, draw and name the mirror images of these compounds.
-
The following four structures are naturally occurring optically active compounds. Star the asymmetric carbon atoms in these structures. CHO H CH, COOH OH OH H,N H serine erythrose menthol camphor
-
Give an example of one piece of financial information that may be used differently for two stakeholders (users) and explain how it would be used and presented differently?
-
Complete common -size statements (balance sheets) using the data below on page 246. You will be required to create a spreadsheet in MS Excel to display the statements (balance sheets).? In a 1 to 2...
-
A sail boat in the Great South Bay travels 2 miles east and then 4 miles south to get to Fire Island. What is its distance and displacement?

Study smarter with the SolutionInn App


