In the circuit of Figure, let E0 = 12.0 V, R = 3.0 ?, and L =
Question:
In the circuit of Figure, let E0 = 12.0 V, R = 3.0 ?, and L = 0.6 H. The switch is closed at time t = 0. At time t = 0.5 s, find
(a) The rate at which the battery supplies power,
(b) The rate of Joule heating, and
(c) The rate at which energy is being stored in the inductor.
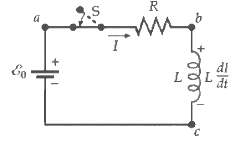
Transcribed Image Text:
Eo a S I R www. IP L dt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a Using Equations 3021 and 3022 We find I 05 s ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
Switch S in Figure is closed at time t = 0, to begin charging an initially uncharged capacitor of capacitance C = 15.0F through a resistor of resistance R = 20.0??. At what time is the potential...
-
In Figure after switch S is closed at time t = 0, the emf of the source is automatically adjusted to maintain a constant current i through S.(a) Find the current through the inductor as a function of...
-
Switch S in Figure is closed at time t = 0 initiating the buildup of current in the 15.0 mH inductor and the 20.0?? resistor. At what time is the emf across the inductor equal to the potential...
-
A machine fills containers with a particular product. The standard deviation of filling weights computed from past data is 0.6 ounces. If only 2% of the containers hold less than 18 ounces, what is...
-
Taylor Equipment Repair Service is owned by Jason Taylor. INSTRUCTIONS Use the following figures to prepare a balance sheet dated February 28, 2019. (You will need to compute the owner's equity.)...
-
Complete and balance the following equations. a. KOH(aq) + MgCl2(aq) b. Mg(s) + CuSO4(aq) c. Sr(s) + H2O(l) d. SrCO3(s) + HCl(aq) e. Ba(OH)2(aq) + CO2(g)
-
When parties must arbitrate their disputes?
-
As an intern with the local home builder's association, you have been asked to analyze the state of the local housing market that has suffered during a recent economic crisis. you have been provided...
-
The declining balance rate is 5 0 % , or ( 2 5 % X 2 ) and the book value at the beginning of the year is $ 3 8 , 0 0 0 . What is the depreciation expense in the second year in dollars?
-
1. Does this grant present Dr. Ray with an ethical dilemma(s) in any way? 2. Derive at least one hypothesis for each research question listed above. Provide a sound rationale or theoretical...
-
The current in a coil with a self-inductance of 1 mH is 2.0 A at t = 0, when the coil is shorted through a resistor. The total resistance of the coil plus the resistor is 10.0 . Find the current...
-
Do Problem 66 for the times t = 1 s and t = 100 s.
-
Suppose you read that 210-dB ultrasound is being used to pulverize cancerous tumors. You calculate the intensity in watts per centimeter squared and find it is unreasonably high (10 5 W/cm 2 ). What...
-
In 2022, the University of Akureyri has recorded the following costs for two administrative units, property management and quality and human resources together with two teaching fields, health...
-
Widgets, Inc., which is not a large corporation, paid income tax in Year 1 in the amount of $35,000. For Year 2, their total income tax is $110,000. They made four timely estimated tax payments for...
-
We generally know that IQ scores are a strong statistical predictor of multiple future life outcomes - income, education levels, health, even longevity. There are hundreds of studies that confirm...
-
You have carried out the below reaction between an acid chloride with an amine to form an amide. Construct a flow chart to show how you would isolate the product from unreacted starting materials...
-
38 Calculate AG for the reaction HO(1) H+(aq) + OH(aq) at 25C for the following conditions: (a) [H] 1.0 107 M, [OH] = 1.0 107 M (b) [H]=1.0 10-3 M, [OH] = 1.0 10 4 M (c) [H] 1.0 10-12 M, [OH] = 2.0...
-
All life on Earth is based on one of many two-polymer systems, each made up of a protein and a nucleic acid. In a cell, these polymers are, for the most part, charge-balanced, with the nucleic acid...
-
In Problems 1522, find the principal needed now to get each amount; that is, find the present value. To get $750 after 2 years at 2.5% compounded quarterly.
-
How would you express 600 in. 1 in.?
-
Repeat Question 22 for the power needed rather than the work.
-
Why is it easier to climb a mountain via a zigzag trail than to climb straight up?
-
Recall from Chapter 4, Example 4-14 that you can use s pulley and ropes to decrease the force needed to raise a heavy load (See Fig. 6-34). But for every meter the load is raised, how much rope must...
-
For its three investment centres, Stahl Company accumulates the following data: Centre I Centre II Centre III Sales $1,966,000 $4,029,000 $3,927,000 Controllable margin 1,096,700 2,145,960 3,858,880...
-
What is a mirror's radius of curvature if cars 1 9 . 0 m away appear 0 . 3 7 their normal size? Express your answer to two significant figures and include the appropriate units.
-
Reading Coming to a new awareness of organizational culture, Edgar H.Schein: https://docs.google.com/document/d/15yKbiEIwu47U1OnyjYevwz9ly42o2PCi0x-Zz_4C-po/edit In the assigned reading from the...
College Oral Communication 1 English For Academic Success 1st Edition - ISBN: 0618230165 - Free Book

Study smarter with the SolutionInn App


