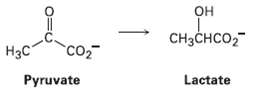
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the
Question:
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the Re face, what is the stereochemistry of theproduct?
Transcribed Image Text:
OH он CH3CHCO2 H3C CO2 Lactate Pyruvate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Strategy Use the strategy in the previous problem to id...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
If lactic acid is the buildup product of strenuous muscle activity, why is sodium lactate often given to hospital patients intra-venously?
-
The pyruvate ( lactate reaction in animals is reversible, but the pyruvate ( ethanol fermentation in yeast is irreversible. Explain.
-
What results from applying Gauss-Jordan reduction to a nonsingular matrix?
-
PIM Industries, Inc., manufactures electronics components. Each unit costs $30 before the final test. The final test rejects, on average, 5 percent of the 50,000 units manufactured per year. The...
-
Rank the magnitudes of these units of thermal energy from greatest to least: a. 1 calorie b. 1 Calorie c. 1 joule
-
Consider a circular DHT with node and key identifiers in the range [0,63]. Suppose there are eight peers with identifiers 0, 8, 16, 24, 32,40,48, and 56. a. Suppose each peer can have one shortcut...
-
AEK Ltd has prepared its income statement, summarised below, for the year ended 30 June 2025. The company is evaluating three independent situations and has asked for your assistance. Required (a) If...
-
The Imaginary Products Co. currently has debt with a market value of $300 million outstanding. The debt consists of 9 percent coupon bonds (semiannual coupon payments) which have a maturity of 15...
-
Use the squeeze theorem to evaluate lim 2 x sin (4x). lim 2 x sin (4x)= Number 0
-
Assume it is now December 31, 2013, and Nicole has just completed her first year of operations at Nicole's Getaway Spa. After looking through her trial balance, she noticed that there are some items...
-
Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2 "CH2 - H Hydroxyacetone Crotyl alcohol
-
The aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or the Si face of the...
-
The energy of a hydrogen atom is \(-3.40 \mathrm{eV}\). What is the electron's kinetic energy? A. \(1.70 \mathrm{eV}\) B. \(2.62 \mathrm{eV}\) C. \(3.40 \mathrm{eV}\) \(\begin{array}{ll}\text { D. }...
-
Inc. settled an overdue account in the amount of $2,000 on January 16 with YYY Company. The customer signed a 30-day promissory note bearing 6% to the lender. What would be the JEs for both companies...
-
Continuing with Bolt data in the previous problem, assume Bolt budgeted a 10.0% share sales of total bolt products in the local market. Reliable industry data indicates that the actual total number...
-
Mary and Joseph are married with 2 children: Jesse, age 1 5 , and Jessica, age 8 . For the current year, the couple s taxable income is $ 3 0 0 , 0 0 0 . What is their tax credit amount related to...
-
Walaa Insurance Company has a surplus reinsurance treaty with Munich Re reinsurance company with retention limit of 1,500,000 SR from the sum insured. The maximum capacity of the surplus treaty is 8...
-
Eddie slick Haskell needs your tax advice regarding a recent stock transaction that didnt turn out the way he planned. On January 20, 2021, Slick purchased 100 shares of Tesla stock (ticker TSLA) for...
-
What are the ethical implications of managing oversharing coworkers? Is sharing too much information about their hobbies, tattoos, religion, food interests, relationships, and children just normal...
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
Summarize each of the simple gas laws (Boyles law, Charless law, and Avogadros law). For each, explain the relationship between the two variables and also state which variables must be kept constant.
-
Draw all enol isomers of the following compounds. If there are none, explain why. (a) 2-methylpentanoic acid (b) Benzaldehyde
-
Draw all enol isomers of the following compounds. If there are none, explain why. (a) 2-methylpentanoic acid (b) Benzaldehyde
-
Crossed aldol condensations can be carried out if one of the carbonyl compounds is unusually acidic. Explain why the aldol condensation of acetone with itself does not compete with the crossed aldol...
-
You are a supervisor at your company. You've overheard Employee A making offensive comments to other employees, and Employee B has complained about these comments. You've given Employee A a verbal...
-
What is the probability of developing and dying from cancer today?
-
On January 1, 2024, Majestic Mantles leased a lathe from Equipment Leasing under a finance lease. Lease payments are made annually. Title does not transfer to the lessee and there is no purchase...

Study smarter with the SolutionInn App


