Look at figure, and tell which molecular orbital is the HOMO and which is the LUMO for
Question:
Look at figure, and tell which molecular orbital is the HOMO and which is the LUMO for both ground and excited states of ethylene and 1, 3-butadiene.
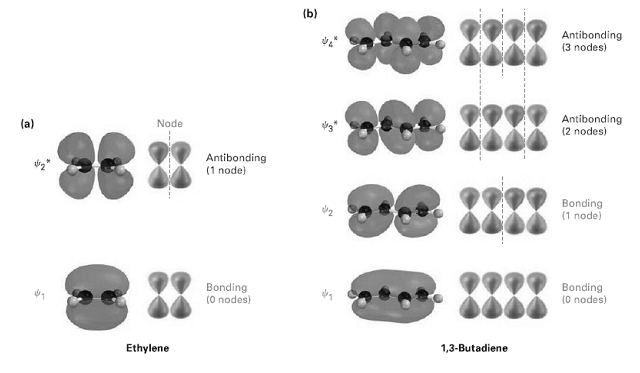
Transcribed Image Text:
(b) Antibonding (3 nodes) Antibonding (2 nodes) (a) Node Antibonding (1 node) Bonding (1 node) Bonding (0 nodes) Bonding (0 nodes) 1,3-Butadiene Ethylene ------- -- -----
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
For ethylene Energy 88 Two 2p atomic orbitals 42 LUMO Antib...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which molecular orbital of ethylene ( or *) is the most important one to look at in a reaction in which ethylene is attacked by an electrophile?
-
Look at the following orbital diagrams and electron configurations. Which are possible and which are not, according to the Pauli exclusion principle? Explain. a. b. c. d. 1s22s22p4 e. 1s22s42p2 f....
-
Ethylene (C2H4) at 25oC and 1 atm is burned with 300% excess air at 25oC and 1 atm. Assuming that this reaction takes place adiabatically at 25oC and that the products leave at 25oC and 1 atm....
-
On January 2, $217981 in 10-year, 5% bonds with a market interest rate of 9%, and interest payable semiannually, were issued for $185895. On June 30, bond interest was paid. On December 31, the...
-
Why did Citibank and Travelers resort to a co-CEO arrangement when they merged in 1998? What are the advantages and disadvantages of such an arrangement?
-
In Example 13.1, assume that the averages of all factors other than educ have remained constant over time and that the average level of education is 12.2 for the 1972 sample and 13.3 in the 1984...
-
In a heat engine the heat always flows from (a) The higher-temperature region to the lower-temperature one and produces no work (b) The lower-temperature region to the higher-temperature one and...
-
The following information, taken from the books of Herman Brothers Manufacturing represents the operations for January: The job cost system is used, and the February cost sheet for Job M45 shows the...
-
what ways do you feel a sense of identity and belonging within the organization's community, and how does that influence your performance and engagement?
-
Calculate the account balance for the following accounts: Service Revenue Accounts Payable Salaries Expense Cash Dr. Cr. Dr. Cr. Dr. Cr. Cr. 9,500 3,200 4,500 1,050 Dr. 6,740 1,720 495 2,500 8,720...
-
The amino acid cysteine, C3H7NO2S, is biosynthesized from a substance called cystathionine by a multistep pathway. (a) The first step is a transamination. What is the product? (b) The second step is...
-
Draw the products you would expect from conrotatory and disrotatory Cyclizations of (2Z, 4Z, 6Z)-2, 4, 6-octatrienc. Which of the Iwo paths would you expect the thermal reaction to follow?
-
Is it accurate to say that central banks have complete control over nominal interest rates? Explain why or why not.
-
3 Dexter Industries purchased packaging equipment on January 8 for $90,000. The equipment was expected to have a useful life of years, or 18,000 operating hours, and a residual value of $3,600. The...
-
Accounts receivable turnover and days' sales in receivables For two recent years, Robinhood Company reported the following: 20Y9 20Y8 Sales $8,113,000 $6,903,000 Accounts receivable: Beginning of...
-
Calculate the yield to maturity of a coupon bond face value $10, 000, purchased for $8000.00 with an interest rate of 8%per annum that has 25 years to maturity and sold at 15 years.
-
Suppose that you purchase a three-year coupon bond with annual coupons of 2%, paid semi-annually and a face value of $100. Suppose that the term structure is flat at 2% APR compounded semi-annually....
-
Valentina is age 40 and her superannuation balance is in the accumulation phase. For the current financial year her investments have produced income returns of $12000 and capital returns of $22000....
-
Rank the following from lowest in the food chain to highest in the food chain: secondary consumer, primary consumer, top predator, producer.
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
High frequency trading is the practice of executing multiple transactions for securities followed by extremely short holding periods. Such market practices often require highly sophisticated...
-
Which of the isomeric alcohols having the molecular formula C5H12O are chiral? Which are achiral?
-
Write structural formulas or make molecular models for all the compounds that are trichloro derivatives of cyclopropane. (Don't forget to include stereoisomers.) Which are chiral? Which are achiral?
-
In each of the following pairs of compounds one is chiral and the other is achiral. Identify each compound as chiral or achiral, as appropriate. a. b. c. d. CICH CHCH OH and HOCH CHCH,OH CI OH CH,CH...
-
x-8x+15 A) Let f(x) = = x2+2x-15 Calculate lim f(x) x 3 2x225x75 B) Let f(x) = x-18x45 Calculate lim f(x) x 15
-
If a dance instructor prices her lessons at $60 per student, she will have three students. If she prices her lessons at $50 per student, she will have four students. How much marginal revenue will...
-
What is the difference between objectives, strategies, and goals? How do these things improve our planning process?

Study smarter with the SolutionInn App


