Name the following aldehydes and ketones according to IUPAC rules: (c) (a) , (b) CCH2 CHCH2CHCH C
Question:
Name the following aldehydes and ketones according to IUPAC rules:
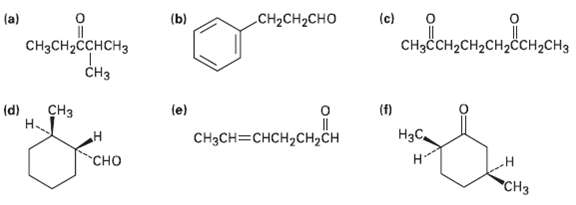
Transcribed Image Text:
(c) (a) сыдоненондсонен, (b) CнаCH2сно CHзCH2CсHCHз CНз CHзCCH2CH2сH2ссH-CHз (e) CHз (f) (d) "opноск CH;CH3CHCH,Cн,ҫH Нас, н н* "сно н "CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Remember that the principal chain must contain the aldehyde or ketone group and that an aldehyde ...View the full answer

Answered By

Joan Gakii
I'm a meticulous professional writer with over five years writing experience. My skill set includes
- Digital Content,
- Interpersonal Communication,
- Web Content and academic Writing,
- Proofreading,
- Editing,
- Project Management, and
- Public Relations.
5.00+
7+ Reviews
12+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name the following molecules according to the IUPAC system of nomenclature. (a) (b) (c) (d) (e) CH3CH(CH3)CH(CH3)CH(CH3)CH(CH3)2 (f) CH3CH2CHCH3 CH CH H3C CH3 CHCHCH2CH3 EH CHCH,CH.CCH,CH.CH CH,...
-
Name the following molecules according to the IUPAC nomenclature system. (a) (b) (c) (d) (e) (f) CH32 Cl CH3 Br
-
Name all the aldehydes and ketones that have the formula C5H10O.
-
What freedom does an international organisation have in regard to imposing its own approaches to HRM on its operations throughout the world? How can an international organisation, aware of the need...
-
1. What are several methods that crooks use to steal your identity? 2. How do you discover that someone has stolen your identity? 3. What steps can you take to thwart identity thieves? 4. What...
-
The Shiners are planning to drive to the Upper Peninsula from Detroit. They arranged to rent a car for $55.75 a day with no charge for mileage. a. What will it cost the Shiners to rent the car for 10...
-
What is the difference between Mission System versus Enabling System CIs? What are their relationships. Using an office building, automobile, and an aircraft, illustrate the relationships.
-
Ted Glickman, the administrator at D. C. General Hospital emergency room, faces the problem of providing treatment for patients who arrive at different rates during the day. There are four doctors...
-
How do advanced frameworks for conflict resolution, such as interest-based negotiation or integrative bargaining, differ from traditional models, and which are most effective in multicultural...
-
Assuming the cost of an associate leaving within 90 days is $3,000, what will be your facility's approximate cost of early turnover for this year? Year-to-Date Turnover Avg. Head- count Total < 90...
-
Repeat Sample Problem39-6 for the Balmer series of the hydrogen atom.
-
Draw structures corresponding to the following names: (a) 3-Methylbutanal (b) 4-Chloro-2-Pentanone (c) Phenyl acetaldehyde (d) cis-3-tert-Butylcyclohexanecarbaldehyde (e) 3-Methyl-3-butenal (f)...
-
Consider a baseband binary PAM system that is designed to have a raised-cosine spectrum P (). The resulting pulse p (t) is defined in Equation (4.62). How would this pulse be modified if the system...
-
The following information is from ABC Company's general ledger: Beginning and ending inventories, respectively, for raw materials were $12,000 and $15,000 and for work in process were $30,000 and...
-
2) At the post office, Tiffany paid $11.04 for 24 stamps. At this rate, how much would it cost for Tiffany to buy 18 stamps? 3) At a factory, there are 40 new cars produced every 8 hours. How many...
-
Find the change in a disk's angular speed (in rad/s) if its mass and radius are 0.230 kg and 0.180 m, respectively
-
Head First Company plans to sell 5, 100 bicycle helmets at $78 each in the coming year Product costs include Direct materials per helmet $32 Direct labor per helmet 5.50 Variable factory overhead per...
-
If f is any function from A into B, we can describe the inverse image as a function from B into P(A), which is also commonly denoted . If b B, (b) = {a A | f(a) = b}. If does have an inverse, the...
-
An analyst stated that a callable bond has less reinvestment risk and more price appreciation potential than an otherwise identical noncallable bond. The analysts statement most likely is a....
-
Why is it important to understand the macro-environment when making decisions about an international retail venture?
-
(A) Refer to Figure 11-26. Write a molecular orbital occupancy diagram, determine the bond order, and write the electronic configurations of (a) N 2 + ; (b) Ne 2 + ; (c) C 2 2- . (B) The bond lengths...
-
The structure of the sex pheromone (attractant) of the female tsetse fly has been confirmed by the following synthesis. Compound C appears to be identical to the natural pheromone in all respects...
-
Provide reagents that would accomplish each of the following syntheses. Begin by writing a retrosynthetic analysis. (a) (b) HO HO or OH
-
Write a detailed mechanism for the following reaction. OH OH H2SO4 (cat), H2O HO OH
-
A 5.0 g bullet moving 325 m/s is shot into a 1.25 kg block, which slides for 1.35 seconds across the surface it is on before coming to rest. What is the average force of kinetic friction between the...
-
Some enterprising physics students working on a catapult decide to have a water balloon fight in the school hallway. The ceiling is of height 3 . 4 m, and the balloons are launched at a velocity of 9...
-
1-Define electric fields and how it helps us understand electricity. 2-Electric fields are represented as a physical effect of a configuration of charges that is created by the attraction of electric...

Study smarter with the SolutionInn App


